Curated OER
Atomic Models
Your students should enjoy this slideshow which includes some basic definitions about the atomic structure of atoms. In fact, this presentation would be best-viewed before giving details and diagrams of neutrons, protons and quarks. The...
Curated OER
Nuclear Reactions - Rutherford's Alpha Scattering Experiment
Neat! Show your physics class exactly how Ernest Rutherford fired alpha particles at a piece of gold foil and so determined that most of an atom is composed of empty space. Not just a slide show, but almost an animation, this...
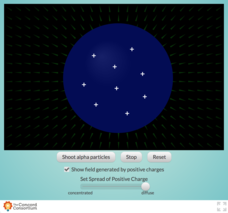
Concord Consortium
Concentrating Charge and Electric Fields
How did Rutherford determine that the nucleus was the center of an atom? Take a look inside the famous Gold Foil Experiment with an interesting interactive. Learners fire a beam of alpha particles at a nucleus containing variable...
Curated OER
Regents High School Examination PHYSICAL SETTING CHEMISTRY 2007
The University of the State of New York has designed a series of exams to be given to high schoolers. This chemistry exam is one of the most comprehensive and well-written that you will ever find. It consists of 84 questions in a variety...
Curated OER
History of the Atom Quiz Review
In this history of the atom worksheet, students answer questions about Thomson's cathode ray apparatus, Rutherford's gold foil experiment and Dalton's beliefs about the atom. Students identify the number of protons, neutrons and...
Curated OER
Understanding the Rutherford Model
In this Rutherford model worksheet, students read about Rutherford's experiment with gold foil and alpha particles which led to the discovery of subatomic particles. Students answer six analysis questions about the experiment and...
Curated OER
Review Models of the Atom
Multiple representations of the varying theories of atomic structure. With these pictures and some background of the theories and scientific figures surrounding them, your class will develop a better understanding of scientific history...
Science Geek
Atomic Structure
The International Union of Pure and Applied Chemistry (IUPAC) was formed in 1919 and was crucial for allowing scientists to discuss findings during the Cold War. A presentation offers an introduction to atomic structure including the...
Virginia Department of Education
Historical Models of Atoms
What does the past have to do with today? Young scientists find that answer as they learn more about past chemists and their significant contributions to the field. Pupils use the Internet to research historical figures and create a...
Curated OER
Atom Review
In this atom worksheet, learners review the subatomic particles, the scientists associated with the atomic theory, and the periodic table. This worksheet has 15 matching, 24 fill in the blank, and 11 short answer questions.
Curated OER
Chemistry
In this chemistry worksheet, students complete 83 multiple choice, short answer and problem solving questions on various chemistry concepts.
Curated OER
Defining the Atom
Words, words, words! This presentation uses plenty of them to define the atom, outline the history of what we know about atoms, and explain atomic and mass numbers. That's all! Informative, but not interesting, this slide show would be...
Curated OER
Isotopes; Molecular Notation; Electron Orbits
In this isotopes, molecular notation, and electron orbits worksheet, students read selections pertaining to isotopes, mass number, molecular notation, electric attraction, and electron orbits. In addition, students complete 11 short...
Curated OER
Isotopes
In this isotopes worksheet, students review mass number, molecular notation, electron attraction, and electron orbits. This worksheet has 5 matching and 5 short answer questions.
Curated OER
Elements
For this elements worksheet, learners determine the atomic number, mass, number, electrons, protons, and neutrons for elements. Students review the electromagnetic spectrum. This worksheet has 1 graphic organizer and 13 multiple choice...
Curated OER
The Bohr-Rutherford Model of the Atom
In this atom model worksheet, students explore how scientists developed the Bohr-Rutherford atom model. Students complete 15 fill in the blank statements.