Hi, what do you want to do?
CK-12 Foundation
Ck 12: Percent Composition and Empirical & Molecular Formulas
[Free Registration/Login may be required to access all resource tools.] In this online tutorial students will learn to calculate the percent composition of a compound either from mass data or from the chemical formula. They will use...
CK-12 Foundation
Ck 12: Plix Series: Percent Composition
[Free Registration/Login Required] Manipulate graph data to see a representation of the percent composition of different compounds. Then answer a challenge question about the activity.
Carnegie Mellon University
Chem Collective: Composition Determination of a Mixture
In this activity, students calculate the percent composition of a mixture of two arsenic-containing minerals. Step-by-step support and feedback is provided for students who need additional help.
Frostburg State University
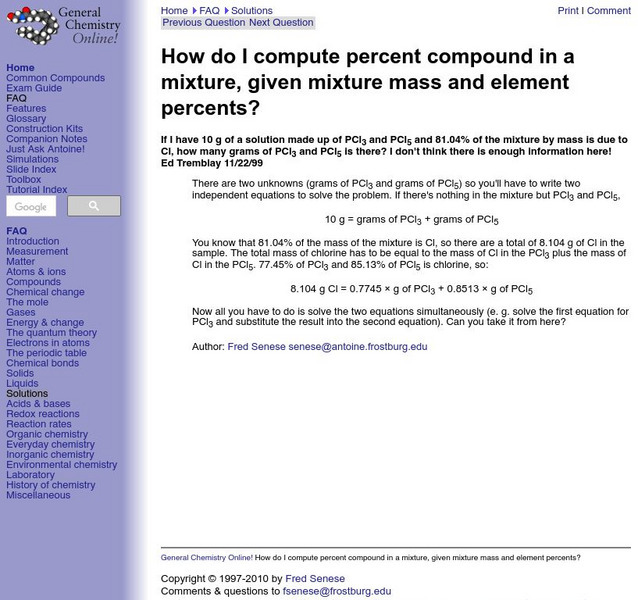
General Chemistry: Percent Compound in a Mixture
Resource contains and example problem and solution of how to compute percent compound within a mixture when given the mixture's mass and element percents.
CK-12 Foundation
Ck 12: Chemical Formulas
[Free Registration/Login may be required to access all resource tools.] Students will use a chemical formula or mass data to calculate the percent composition of a compound, and then calculate the empirical or molecular formula for a...
Carnegie Mellon University
Chem Collective: Mineral Composition
In this randomized calculation activity, students calculate the empirical formula of a compound given its elemental analysis. Step-by-step support and feedback is provided for students who need additional help.
Science Struck
Science Struck: Composition of Air
Discover all the elements and chemical compounds that are found in the air. They are presented in a chart listing their symbols, molecular weights, and what percent of the atmosphere they each comprise. A set of facts is also provided.
Clackamas Community College
Clackamas Community College: Empirical Formulas
This site presents as explanation for calculating empirical formulas from composition data. Numerous examples are given, along with practice problems.
James Madison University
James Madison University: Igneous Rock Classification
This site explores the classification of igneous rocks. Content addresses classification based on color and texture, mineral composition, and chemical composition.
Texas Education Agency
Texas Gateway: Empirical Formula
This tutorial reviews how to calculate molecular formula by giving students video, exercises, and practice.
Upper Canada District School Board
Tom Stretton's Advanced Placement Chemistry: Stoichiometry
This online textbook chapter provides learners with advanced-level reading and practice material on stoichiometry
Towson University
Towson University: Determining Formulae
Explanation of how to calculate empirical and molecular formulas.
CK-12 Foundation
Ck 12: Chemistry Simulation: Average Atomic Mass
[Free Registration/Login Required] Students explore how percent abundances of different isotopes affect the average atomic mass for that given element. Students form connections between the average atomic mass and a person's body mass....
Other popular searches
- Percent Composition Lab
- Percent Composition by Mass
- Percent Composition Quiz
- Mass Percent Composition