Hi, what do you want to do?
CK-12 Foundation
Ck 12: Limiting Reactant and Percent Yield
[Free Registration/Login may be required to access all resource tools.] In the following online tutorial students wil begin to analyze a chemical reaction in order to determine which reactant is the limiting reactant and which is the...
Khan Academy
Khan Academy: Limiting Reagents and Percent Yield
How to determine the limiting reagent, and using stoichiometry to calculate the theoretical and percent yield.
CK-12 Foundation
Ck 12: Limiting Reactant and Percent Yield
[Free Registration/Login may be required to access all resource tools.] Students will compare theoretical yield to actual yield, and then investigate what happens when one reactant runs out before the other reactants are fully consumed....
Chiral Publishing
Chiral Publishing: An Introduction to Chemistry: Real World Applications of Equation Stoichiometry
This audio book, narrated by author Mark Bishop, describes how equation stoichiometry can be used in the real world. Many examples are given to help understand how to plan chemical reactions, solve for limiting reactants, and calculate...
Frostburg State University
General Chemistry Online: The Mole Concept
Resource provides notes on Moles and Stoichiometry. Deals with all book-keeping aspects, including as section on yields and limiting reactants. Includes lesson plans, lecture slides and notes, links to related websites, and frequently...
Frostburg State University
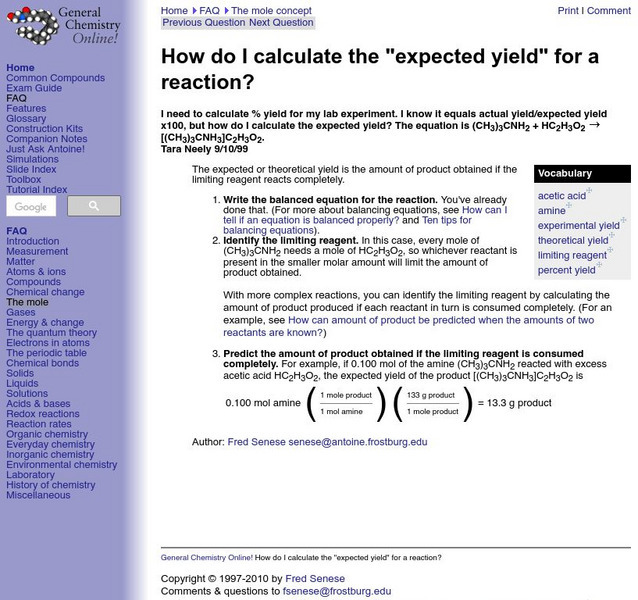
General Chemistry Online: Expected Yield
Frostburg State University provides an example of a problem dealing with how to find expected yield. Includes step-by-step directions on how to solve the problem.
Science Education Resource Center at Carleton College
Serc: Molar Relationships: Observing a Single Displacement Reaction
In this chemistry lab, learners will investigate a single replacement reaction while also using knowledge of molar ratios to determine percent yields.