Hi, what do you want to do?
University of Colorado
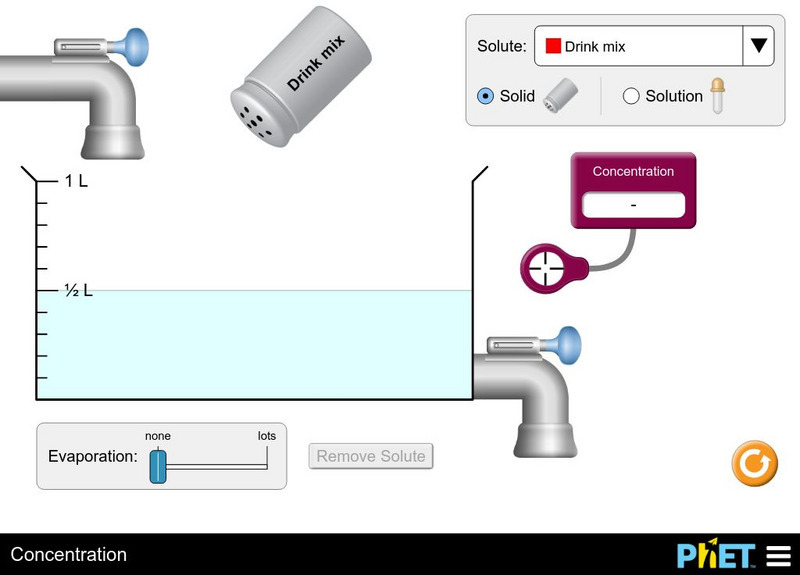
University of Colorado: Ph Et Interactive Simulations: Concentration
Watch your solution change color as you mix chemicals with water. Then check molarity with the concentration meter. What are all the ways you can change the concentration of your solution? Switch solutes to compare different chemicals...
University of Colorado
University of Colorado: Ph Et Interactive Simulations: Concentration
Learn how concentration, evaporation, and saturation affect the concentration of a solution.
Utah Education Network
Uen: Fat a Concentrated Energy Source
Through these lesson plans, students explore and discuss the role of dietary fat as an important energy source. In addition to providing detailed instructions for teachers, this website includes all the handouts and charts needed to...
University of Colorado
University of Colorado: Ph Et Interactive Simulations: Molarity
What determines the concentration of a solution? Learn about the relationships between moles, liters, and molarity by adjusting the amount of solute and solution volume. Change solutes to compare different chemical compounds in water.
Chem Tutor
Chem Tutor: Dissolving Gases Into Liquids
Description of the factors influencing gas solubility. This site also provides other chemistry related questions. Topics include dissolving solids into liquids, concentration, and stoichiometry.
Ducksters
Ducksters: Chemistry for Kids: Solutions and Dissolving
On this site, students learn about solutions and dissolving in chemistry including interesting facts, examples, solubility, saturation, concentration, and what is a solution?
ClassFlow
Class Flow: Solubility and Solution
[Free Registration/Login Required] This flipchart explains the concepts of solubility, saturated, unsaturated, supersaturated, the factors affecting solubility, and molarity concentration calculations.