Hi, what do you want to do?
Davidson College
Davidson College: Valence Shell Electron Pair Repulsion (Vsepr) Model
Presents examples of molecules that do not match the expected bond angles in the Valence-Shell Electron-Pair Repulsion Model. Requires Java.
Davidson College
Davidson College: Valence Shell Electron Pair Repulsion (Vsepr) Model
Explains the principles of the Valence-Shell Electron-Pair Repulsion Model, and gives examples of the geometry. Requires Java.
Davidson College
Davidson College: Valence Shell Electron Pair Repulsion (Vsepr) Model
Presents exercises where students can check their understanding of the Valence-Shell Electron-Pair Repulsion Model by predicting what a molecular shape will be. Requires Java.
University of Waterloo (Canada)
University of Waterloo: Vsepr Models
The University of Waterloo provides background in pair repulsion and confidence building questions. Try this one once you think you know what you are doing!
Other
Us: Valence Shell Electron Pair Repulsion (Vsepr)
An excellent tutorial that examines VSEPR and pairs of valence electrons. The valence shell electron pair repulsion concept is explored using animated models. Includes a VSEPR calculator. Use the toolbar on the left to navigate through...
CK-12 Foundation
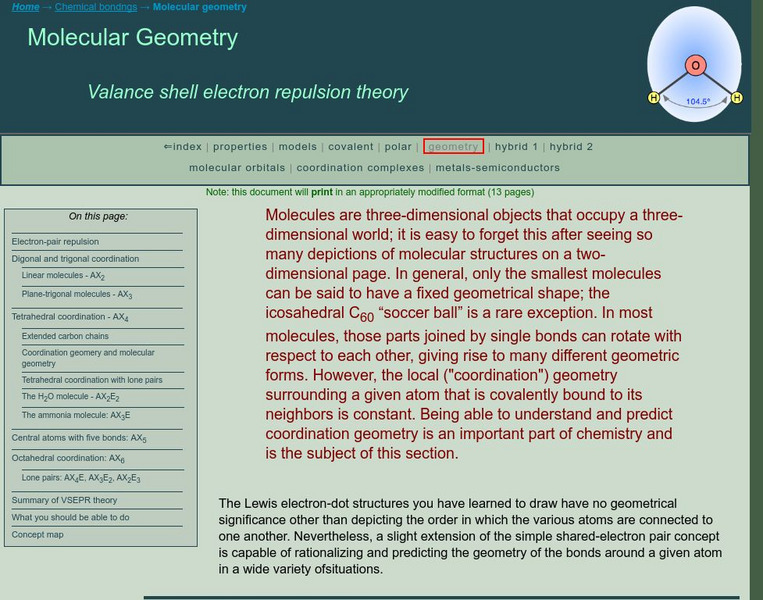
Ck 12: Molecular Geometry
[Free Registration/Login may be required to access all resource tools.] The following online tutorial explains the basis of VSEPR theory. It helps students predict the shapes of molecules and polyatomic ions using VSEPR theory and it...
Simon Fraser University
Chem1 Virtual Textbook: Molecular Geometry
An advanced explanation of the valence shell electron pair repulsion (VSEPR) theory describes specific molecular models involving digonal, trigonal, tetrahedral, and octahedral coordination, as well as central atoms with five bonds....
University of Colorado
University of Colorado: Ph Et Interactive Simulations: Molecule Shapes
Explore molecule shapes by building molecules in 3D. How does molecule shape change with different numbers of bonds and electron pairs? Find out by adding single, double or triple bonds and lone pairs to the central atom. Then, compare...