Hi, what do you want to do?
OpenStax
Open Stax: Andrew R. Barron: Valence Shell Electron Pair Repulsion (Vsepr) Theory
Detailed explanation of Valence Shell Electron Pair Repulsion (VSEPR) Theory with examples and questions for the reader to check comprehension. Also includes step-by-step instructions for correctly building molecules using VSEPR Theory....
PBS
Pbs Learning Media: Molecular Shapes
In this interactive activity from ChemThink, students will learn about covalent molecules and how the VSEPR theory predicts the shapes of covalently-bonded molecules.
CK-12 Foundation
Ck 12: Molecular Geometry
[Free Registration/Login may be required to access all resource tools.] The following online tutorial explains the basis of VSEPR theory. It helps students predict the shapes of molecules and polyatomic ions using VSEPR theory and it...
Simon Fraser University
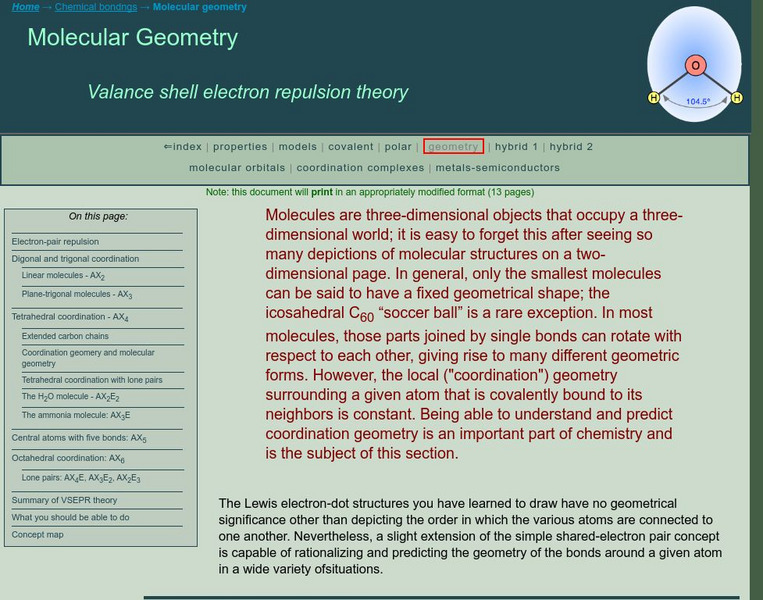
Chem1 Virtual Textbook: Molecular Geometry
An advanced explanation of the valence shell electron pair repulsion (VSEPR) theory describes specific molecular models involving digonal, trigonal, tetrahedral, and octahedral coordination, as well as central atoms with five bonds....
Chem Tutor
Chem Tutor: Chemistry: Compounds
This lesson focuses on chemical compounds including Ionic and Covalent Bonds, Valences, Lewis Structures, Binary Covalent Compounds, Radicals or Polyatomic Ions, and much more. It also includes a compound worksheet in which the students...
CK-12 Foundation
Ck 12: Molecular Geometry
[Free Registration/Login may be required to access all resource tools.] In this interactive learning module, students will learn a technique to predict molecular geometry based on a molecule's Lewis electron dot structure.
Towson University
Towson University: Shapes of Molecules
This chemistry class printout details the main points of molecular geometry and explains bond hybridization and bond angles.