Hi, what do you want to do?
Environmental Chemistry
Environmental chemistry.com: Periodic Table Hassium
As the name of this site implies, this is a huge reference site for elemental information. Whether you are looking for properties or isotopes, this site probably has what you need. Images of structure and electron energy level can be...
Purdue University
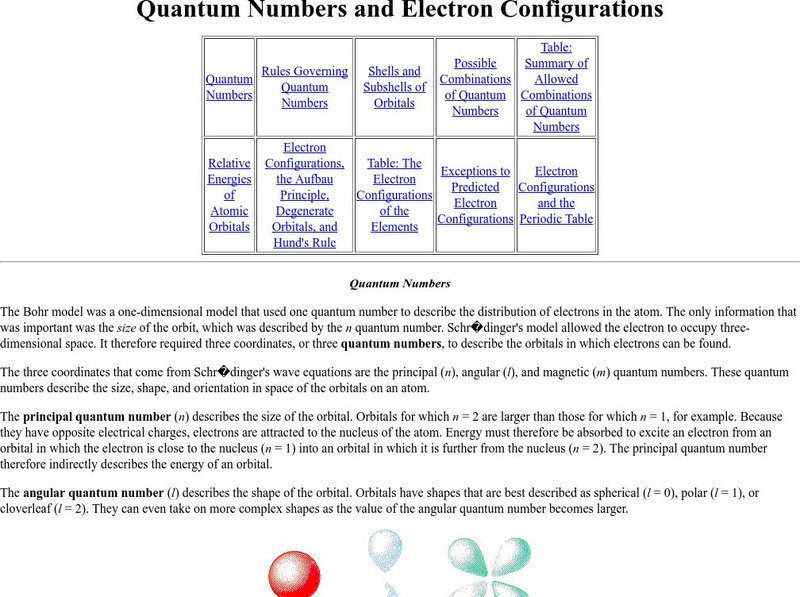
Purdue University: Quantum Numbers & Electron Configurations
This site from the Purdue University provides quantum numbers explained in detail and shown how they specify the atomic orbital of each electron and their energy levels. Electronic configurations are explained. Learning exercises with...
University of St. Andrews (UK)
University of St. Andrews: Insulators
The nature of insulators is described at the atomic level. Band gap theory is used to explain what distinguishes insulators from conductors.
University of Colorado
University of Colorado: Physics 2000: Spectral Lines
Several pages from an excellent site which describe the science of spectroscopy. The unique atomic emission (and absorption) line spectrum of elements are illustrated and explained. Includes a Java applet depicting the quantum energy...
Michigan State University
Michigan State University: Elementary Physics Ii: The Pauli Exclusion Principle
The Pauli Exclusion Principle requires that all electrons in an atom have unique energy levels.
Chem4kids
Chem4 Kids: Atoms: Orbitals
The website provides an explanation of the electrons' position in their orbitals as they spin around the nucleus. Quantum theory is also explored by explaining that the exact location of a specific electron is a not exact but a guess.
American Chemical Society
Middle School Chemistry: Represent Bonding With Lewis Dot Diagrams
Students draw and interpret Lewis dot diagrams for individual atoms and both covalent and ionic compounds.
Friesian School
Proceedings of the Friesian School/the Quantacized Atom
A very lengthy page from friesian.com discussing Bohr's theory of electronic energy levels and the explanation of commonly observed atomic emission line spectra. The concept of a photon and Einstein's observation of the photoelectric...
Other
Chemical Elements: Potassium
A nice, clear site, containing a good deal of elemental information, including atomic weight, density, boiling point, isotopes and more. An image shows the electron energy levels for the element.
Other
Chemical Elements: Plutonium
A nice, clear site, containing a good deal of elemental information, including; atomic weight, density, boiling point, isotopes and more. An image shows the electron energy levels for the element.
Dartmouth College
Dartmouth College: Chem Lab: Electrons in a Box
This site has an interactive component that allows you to change the mass, number of electrons and the length of the box and see how it affects the energy levels. Requires Java.
University of Kentucky
University of Kentucky: Hydrogen Like Orbitals
Good site for viewing atomic orbitals. Click on an energy level and you get a simulated three dimensional image of the orbitals.
Curated OER
Proceedings of the Friesian School/the Quantacized Atom
A very lengthy page from friesian.com discussing Bohr's theory of electronic energy levels and the explanation of commonly observed atomic emission line spectra. The concept of a photon and Einstein's observation of the photoelectric...
Curated OER
Proceedings of the Friesian School/the Quantacized Atom
A very lengthy page from friesian.com discussing Bohr's theory of electronic energy levels and the explanation of commonly observed atomic emission line spectra. The concept of a photon and Einstein's observation of the photoelectric...
Curated OER
Proceedings of the Friesian School/the Quantacized Atom
A very lengthy page from friesian.com discussing Bohr's theory of electronic energy levels and the explanation of commonly observed atomic emission line spectra. The concept of a photon and Einstein's observation of the photoelectric...
Curated OER
Proceedings of the Friesian School/the Quantacized Atom
A very lengthy page from friesian.com discussing Bohr's theory of electronic energy levels and the explanation of commonly observed atomic emission line spectra. The concept of a photon and Einstein's observation of the photoelectric...
Curated OER
Proceedings of the Friesian School/the Quantacized Atom
A very lengthy page from friesian.com discussing Bohr's theory of electronic energy levels and the explanation of commonly observed atomic emission line spectra. The concept of a photon and Einstein's observation of the photoelectric...
Curated OER
Proceedings of the Friesian School/the Quantacized Atom
A very lengthy page from friesian.com discussing Bohr's theory of electronic energy levels and the explanation of commonly observed atomic emission line spectra. The concept of a photon and Einstein's observation of the photoelectric...
Curated OER
Proceedings of the Friesian School/the Quantacized Atom
A very lengthy page from friesian.com discussing Bohr's theory of electronic energy levels and the explanation of commonly observed atomic emission line spectra. The concept of a photon and Einstein's observation of the photoelectric...
Curated OER
Proceedings of the Friesian School/the Quantacized Atom
A very lengthy page from friesian.com discussing Bohr's theory of electronic energy levels and the explanation of commonly observed atomic emission line spectra. The concept of a photon and Einstein's observation of the photoelectric...
Other
Chemtopics: Understanding the Schrodinger Eqn.
The Schrodinger equation specifies atomic orbitals which are occupied by an electron. Quantum numbers can identify a unique energy level for each electron (set up in a PowerPoint presentation).
Curated OER
[Bohr Model of Holmium]
A nice, clear site, containing a good deal of elemental information about the element holmium, including atomic weight, density, boiling point, isotopes and more. An image shows the electron energy levels for the element.
Curated OER
[Bohr Model of Potassium]
A nice, clear site, containing a good deal of elemental information, including atomic weight, density, boiling point, isotopes and more. An image shows the electron energy levels for the element.
Curated OER
[Bohr Model of Plutonium]
A nice, clear site, containing a good deal of elemental information, including; atomic weight, density, boiling point, isotopes and more. An image shows the electron energy levels for the element.