Hi, what do you want to do?
CK-12 Foundation
Ck 12: Orbitals
[Free Registration/Login may be required to access all resource tools.] A collection of learning opportunities about electron orbitals. Includes videos, activities, discussion questions, and quizzes.
University of Kentucky
University of Kentucky: Hydrogen Like Orbitals
Good site for viewing atomic orbitals. Click on an energy level and you get a simulated three dimensional image of the orbitals.
Concord Consortium
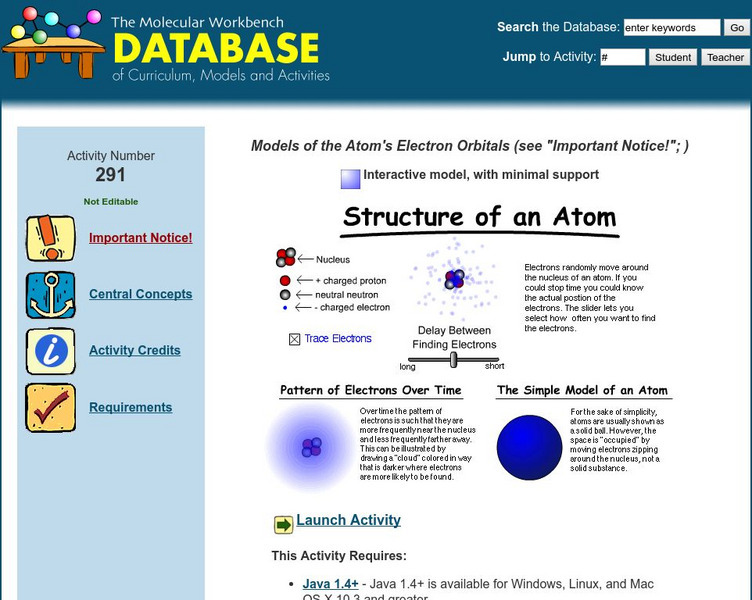
The Molecular Workbench Database: Models of the Atom's Electron Orbitals
Learn about atomic structure and the multiple theories of atomic structure in this simulation.
Vision Learning
Visionlearning: Atomic Theory: Bohr and the Beginnings of Quantum Theory
Description of the ideas and experimentation that led to quantum theory. Focus on electrically charged ions and isotopes.
University of Colorado
University of Colorado: Physics 2000: Elements as Atoms: Electron Clouds and Energy Levels
An explanation of the different types of atomic orbitals, how they are filled according to the Pauli Exclusion Principle, and how many electrons can fit in each electron shell.
University of Southern California
Atomic Bonds
This slide show on atomic bonds contains several slides on electron affinity. Other topics include covalent, Sigma and Pi bonds, and atomic bonding in solids.
Davidson College
Davidson College: Effective Nuclear Charge
Explains what Slater rules are and presents exercises for exploring how they are used to estimate the effective nuclear charge and how well they work compared to the Bohr model expression for orbital sizes. Requires Java.
Chiral Publishing
Chiral Publishing: An Introduction to Chemistry: Multi Electron Atoms: Audio Book
This audio book, narrated by author Mark Bishop, describes how to write regular and abbreviated electron configurations, and how to draw orbital diagrams. Also find links to animations, tutorials, power points, and chapter maps about...
Davidson College
Davidson College: Effective Nuclear Charge
Explains what the effective nuclear charge is and how electrons can create a shield between a nucleus and an outer electron. Presents exercises for comparing orbital sizes with effective nuclear charges. Requires Java.
Simon Fraser University
Chem1 Virtual Textbook: What Is an Orbital?
Acting as part of an overview on quantum theory, this section of the site deals with electrons and orbitals. In addition to explain what an orbital is, the site explains the movement of the electron in relation to the nucleus.
CK-12 Foundation
Ck 12: Plix: Energy Levels: Bohr's Atomic Model
[Free Registration/Login Required] In this interactive you will excite an electron by dragging it into a higher orbital and use the table to see what wavelength of electromagnetic radiation is emitted when it returns to its lowest orbit....
Other
Iun: Modern Atomic Theory
This is an excellent site with information on the discovery of the atom and the different models. Includes a sample question and answer using Planck's constant.
CK-12 Foundation
Ck 12: Modern Atomic Theory
[Free Registration/Login may be required to access all resource tools.] Rutherford's model of the atom was better than earlier models. But it wasn't the last word. Danish physicist Niels Bohr created a more accurate and useful model....
Science Education Resource Center at Carleton College
Serc: Quantum Atomic Structure
This multi-day lesson plan helps students to understand how the models of the atom have changed and how quantum mechanics affects the electrons as they orbit the nucleus.
University of Colorado
University of Colorado: Physics 2000: Elements as Atoms: The Pauli Exclusion Principle
The Pauli Exclusion Principle shows how electrons fill atomic orbitals. Includes biographical information on Wolfgang Pauli.
University of Colorado
University of Colorado: Physics 2000: Elements as Atoms: Quantum Numbers
Each electron has a set of quantum numbers that specify it's location, orbital, and energy in a unique manner.
Other
University of Texas: Tabled Discussion
At this University of Texas site, atomic orbital occupancy, quantum numbers, the Aufbau Principle, and periodic trends are described in detail.
ClassFlow
Class Flow: Atomic Model and Orbital Quiz
[Free Registration/Login Required] This flipchart contains a 20 point multiple choice quiz on atomic history and modeling.
Chiral Publishing
Chiral Publishing: An Introduction to Chemistry: Modern Atomic Theory: Power Point [Pdf]
This power point highlights the main ideas about modern atomic theory from the online textbook "An Introduction to Chemistry". Step by step instructions are given to write electron configurations and draw orbital diagrams. Also includes...
Environmental Chemistry
Environmental chemistry.com: Anatomy of an Atom
Explains the basics of atomic structure, from simple definitions to information about quantum theory. Accurate and helpful basics whether or not you need the more advanced information.
State University of New York
State University of New York: Atomic Electron Configuration
This simulation displays the electron configurations of for the elements in relation to the element's position on the periodic table.
Vision Learning
Visionlearning: Atomic and Ionic Structure of First 12 Elements
Analyze the electron configuration of the of the first twelve elements on the Periodic Table.
Nobel Media AB
The Nobel Prize: Erwin Schrodinger Biographical
This biography from the The Nobel Foundation overviews the life and scientific worlk of Erwin Schrodinger, an Austrian scientist who was honored for his work with atomic theory. Read about his education and personal life, and view links...
Georgia State University
Georgia State University: Hyper Physics: Quantum Numbers, Hydrogen Atom
This tutorial contains links to explanations of the four different quantum numbers (principal, orbital, magnetic, and spin). Equations for each are provided.