Hi, what do you want to do?
American Chemical Society
Middle School Chemistry: Energy Levels, Electrons, and Ionic Bonding
Explore ionic molecular bonds and how electrons and their energy levels come into play.
CK-12 Foundation
Ck 12: Physical Science: Energy Level
[Free Registration/Login may be required to access all resource tools.] Covers energy levels of atoms, their relation to orbitals, and the significance of electrons at different energy levels.
American Chemical Society
Middle School Chemistry: Energy Levels, Electrons, and Covalent Bonding
Students discover the concept that two atoms can attract and form a covalent bond.
Michael Blaber, PhD
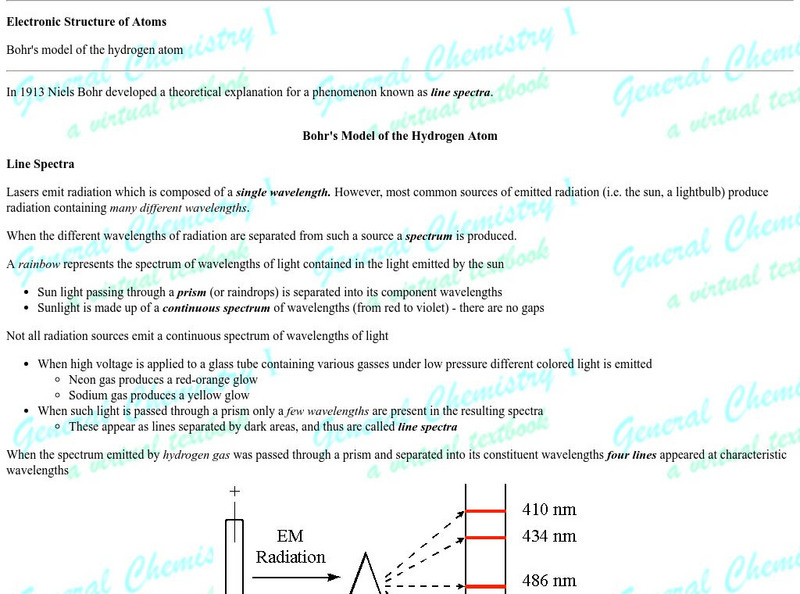
Florida State University: The Bohr Model of the Atom
A well designed clear tutorial explaining the energies involved in the Bohr model of the atom. Illustrations add to the clearly presented equations.
University of Colorado
University of Colorado: Physics 2000: Elements as Atoms: Electron Clouds and Energy Levels
An explanation of the different types of atomic orbitals, how they are filled according to the Pauli Exclusion Principle, and how many electrons can fit in each electron shell.
Science Education Resource Center at Carleton College
Serc: Electron Energy Levels of Atoms and Ions
In this lab, students investigate basic electron structure by making a model using pennies and different sized filter papers.
Other
Ap Physics Lab: Energy Levels of the Hydrogen Atom
A lab activity from an AP Physics course. Students measure the energy changes associated with electron level transitions to the second energy level for hydrogen gas. Includes directions and suggestions. Ideal for a student project or lab...
Concord Consortium
Concord Consortium: Stem Resources: Atoms and Energy
Online activity walks learner through exploration of kinetic and potential energy using animations. Through graphs, a quantitative connection is made between the movement and the energy involved. This information is then related to the...
Walter Fendt
Walter Fendt: Bohr's Theory of the Hydrogen Atom
An explanation of Niels Bohr's (1885 - 1962 CE) Model along with an app that illustrates a hydrogen atom according to the particle or wave model. You can choose a principal quantum number "n." The right part of the graphic represents the...
Simon Fraser University
Chem1 Virtual Textbook: Molecules and the Properties of Bonded Atoms
College-level site goes into great detail about chemical bonding properties, as far as energies, structure, angles, and length. Infrared absorption, the greenhouse effect, and global warming are addressed to exemplify bond length...
American Chemical Society
Middle School Chemistry: Energy Levels, Electrons, Ionic Bonding
Young scholars discover that ionic bonding occurs when electrons are transferred from one atom to the other and not shared as in covalent bonding.
Environmental Chemistry
Environmental chemistry.com: Anatomy of an Atom
Explains the basics of atomic structure, from simple definitions to information about quantum theory. Accurate and helpful basics whether or not you need the more advanced information.
University of Colorado
University of Colorado: Physics 2000: Bohr's Atom
An outstanding site that can best be described as student friendly. It describes the main differences between the classical and quantum models of the atom. Clear and well illustrated.
Concord Consortium
Concord Consortium: Stem Resources: Spectroscopy
Web-based activity walks learners through the use of spectroscopy in determining the identity of atoms and molecules by defining the excited state of atoms, the frequencies of emitted photons, and atoms' ability to absorb them....
Other
Brockport High School: Energy Levels of Hydrogen Atom
From the Brockport High School Physics Labs web pages. Includes an excellent graphic depicting the energy levels of a hydrogen atoms and portraying the electron level transitions for the Lyman, Balmer, and Paschen series. Includes both...
Chem4kids
Chem4 Kids: Atoms: Orbitals
The website provides an explanation of the electrons' position in their orbitals as they spin around the nucleus. Quantum theory is also explored by explaining that the exact location of a specific electron is a not exact but a guess.
University of St. Andrews (UK)
University of St. Andrews: The Quantum Age Begins
This University of St. Andrews page provides an interesting discussion of the beginning of the age of quantum physics. (Quantum physics/mechanics is thought to govern the energy levels of electrons in atoms.) There is a references link...
Georgia State University
Georgia State University: Hyper Physics: Hydrogen Energies and Spectrum
This site from Georgia State University gives information on the transitions of electrons between energy levels. The energy levels for electrons in the hydrogen atom are discussed. The Rydberg equation is stated and electron transitions...
BBC
Bbc: Gcse Bitesize: What Does the Periodic Table Tell Us About the Elements?
The number of protons in the atom of an element determines its place in the Periodic Table. The number of electrons in an atom is the same as the number of protons. These electrons are arranged in shells or 'energy levels' around the...
Friesian School
Proceedings of the Friesian School/the Quantacized Atom
A very lengthy page from friesian.com discussing Bohr's theory of electronic energy levels and the explanation of commonly observed atomic emission line spectra. The concept of a photon and Einstein's observation of the photoelectric...
Michael Blaber, PhD
Fsu: Electronic Structure of Atoms: Quantum Mechanics & Atomic Orbitals
This article is provided by Florida State University. Schroedinger wave equation is explained and how quantum numbers are used to describe the energy level of any electron in an atom. The relationship of atomic orbitals to quantum...
American Chemical Society
Middle School Chemistry: Represent Bonding With Lewis Dot Diagrams
Learners draw and interpret Lewis dot diagrams for individual atoms and both covalent and ionic compounds.
CK-12 Foundation
Ck 12: Heat
[Free Registration/Login may be required to access all resource tools.] In this online lesson students will describe how the macroscopic properties of a thermodynamic system such as temperature, specific heat, and pressure are related to...
American Chemical Society
Middle School Chemistry: Moving Molecules in a Solid
Students observe and describe, on the molecular level, how heating and cooling affect the motion of molecules in a solid.