Curated OER
History of Atomic Theory
Students study the significance of the quantum model and how scientific theories adapt over time. In this investigative lesson students describe the contributions that scientists have helped develop the atomic theory and identify...
Curated OER
Atoms : A Self Guided Computer Activity
"Self-guided Computer Activity" simply means that young chemists read through the slides and take notes about atoms along the way. There is an abundance of text on each slide, making this a comprehensive introduction to atomic structure....
Curated OER
Using several learning modalities to teach about the Periodic Table.
Students identify how to relate the position of an element in the periodic table to its atomic number and atomic mass. They identify how to use the periodic table to identify metals, semimetals, nonmetals, and halogens, and also,...
Curated OER
Atomic Structure and Quantum Theory
Students are introduced to the structure of an atom and Dalton's atomic theory through a short video and mini-lecture. They also take a look at the weight of mass and become familiar with the idea of quantum theory
Khan Academy
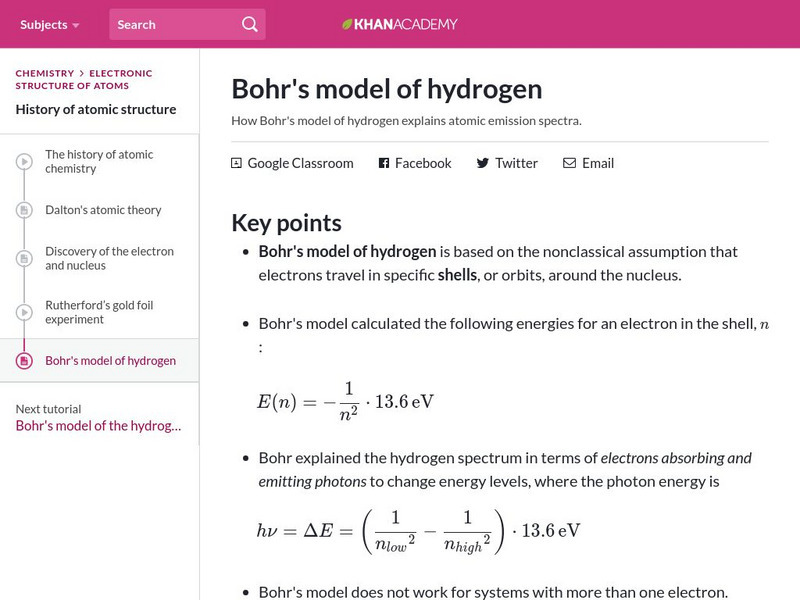
Khan Academy: Bohr's Model of Hydrogen
Resource investigates how Bohr's model of hydrogen explains atomic emission spectra.
Michael Blaber, PhD
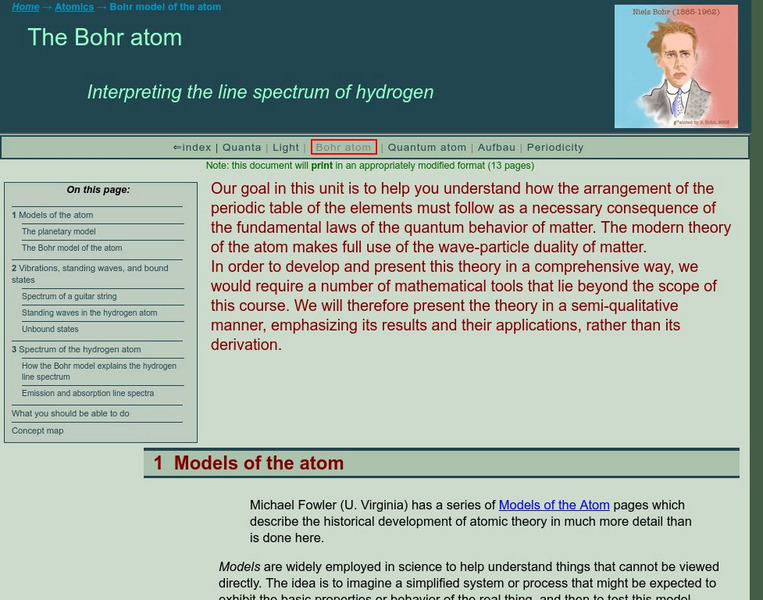
Florida State University: The Bohr Model of the Atom
A well designed clear tutorial explaining the energies involved in the Bohr model of the atom. Illustrations add to the clearly presented equations.
Simon Fraser University
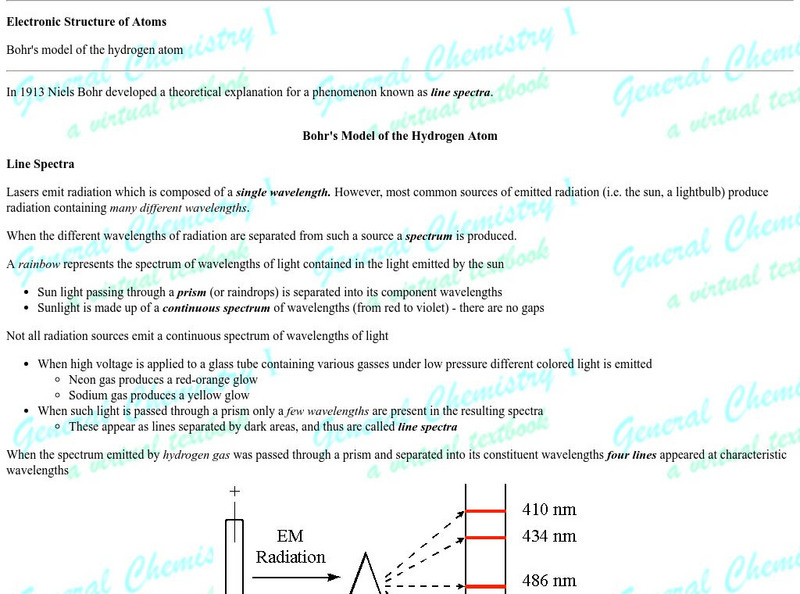
Chem1 Virtual Textbook: Bohr's Model
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses Niels Bohr and his work with the atom. This part of the discussion, focusing on the model itself, brings up...
CK-12 Foundation
Ck 12: The Bohr and Quantum Models of the Atom
[Free Registration/Login may be required to access all resource tools.] Students explore how the study of the hydrogen emission spectrum led to the Bohr model of the atom, in which electrons exist in states of constant energy.
CK-12 Foundation
Ck 12: Flex Book Textbooks: Chemistry Second Edition
[Free Registration/Login may be required to access all resource tools.] A complete, web-based, multi-media textbook covering a wide variety of Chemistry concepts.
CK-12 Foundation
Ck 12: Chemistry: Bohr's Atomic Model
[Free Registration/Login may be required to access all resource tools.] Explains the basic principles of the Bohr hydrogen atom.
Other
Erik's Chemistry Page: Electronic Structure of Atoms
A description of quantum theory, the Bohr model of the atom, the quantum mechanical atom, the Scrodinger equation, and quantum numbers.
Simon Fraser University
Chem1 Virtual Textbook: The Bohr Atom
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses Niels Bohr and his work with the atom. Topics covered in the discussion include the atom before Bohr, Bohr's...
Simon Fraser University
Chem1 Virtual Textbook: Atoms and the Periodic Table
As part of the General Chemistry Virtual Textbook, this site examines a list of related topics (in hyperlink format) on the atom and the Periodic Table of elements. Topics from light, particles, and waves to the Bohr model, quantum...
Other
Crocodile Clips: Absorb Chemistry: History of the Atom
A tutorial that presents models of the atom proposed by John Dalton, J.J. Thomson, Ernest Rutherford, and Niels Bohr. Each is supported by an animated illustration. Includes comprehension questions and a quiz at the end.
Simon Fraser University
Chem1 Virtual Textbook: Bohr's Theory
Acting as part of an overview on quantum theory, this section of the site answers the question, "How did Bohr's theory save the planetary model, for a while?" A section below also discusses the primary problems with Bohr's theory.
Walter Fendt
Walter Fendt: Bohr's Theory of the Hydrogen Atom
An explanation of Niels Bohr's (1885 - 1962 CE) Model along with an app that illustrates a hydrogen atom according to the particle or wave model. You can choose a principal quantum number "n." The right part of the graphic represents the...
Upper Canada District School Board
Tom Stretton's Advanced Placement Chemistry: Atomic Structure and Periodicity
This chemistry e-textbook provides students with AP-level reading and practice material on atomic structure and periodicity.
Simon Fraser University
Chem1 Virtual Textbook: Spectrum of the Hydrogen Atom
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses the hydrogen atom and its relation to spectrum. Included in the discussion is information on the Bohr model...
Davidson College
Davidson College: Effective Nuclear Charge
Explains what Slater rules are and presents exercises for exploring how they are used to estimate the effective nuclear charge and how well they work compared to the Bohr model expression for orbital sizes. Requires Java.
CK-12 Foundation
Ck 12: Chemistry Simulation: Neon Lights
[Free Registration/Login Required] Neon lights are a type of discharge tube. Observe how electrons create colored light in a hydrogen gas discharge tube. Can you figure out why hydrogen's emission spectrum contains more than one color of...
Simon Fraser University
Chem1 Virtual Textbook: Spectrum of a Guitar String
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site discusses spectrum in relation to Bohr's model. Included in the topics covered are standing waves, boundary condition,...
Simon Fraser University
Chem1 Virtual Textbook: Emission and Absorption Spectra
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site specifically discusses emission and absorption spectra in relation to the Bohr model.
CK-12 Foundation
Ck 12: Atomic Theory
[Free Registration/Login may be required to access all resource tools.] In this online tutorial students will explain the law of conservation of mass, the law of definite proportions, and the law of multiple proportions. They will also...
Khan Academy
Khan Academy: The Periodic Table, Electron Shells, and Orbitals
The Bohr model and atomic orbitals. Using an element's position in the periodic table to predict its properties, electron configuration, and reactivity.