Curated OER
Naming More Compounds
In this naming compounds worksheet, students are given the chemical formulas of sixty ionic and covalent compounds and they name each.
CK-12 Foundation
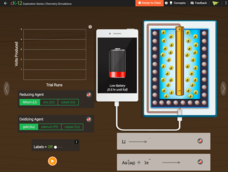
Battery
Don't take for granted the technology behind power packs. Build an understanding of the chemical mechanics of a battery pack that charges your phone. The simulation allows young scientists to manipulate the type of elements in a pack and...
Curated OER
Ionic Chemical Formulas Days 1 & 2
Students study polyatomic ions and write binary and ternary ionic chemical formulas. They explore putting together different monatomic ions and coming up with as many chemical formulas as possible. They play a game similar to memory...
Curated OER
IPC Chemistry Review
In this chemistry review activity, students classify and name compounds, calculate atomic mass, draw Lewis Dot Diagrams, and determine number of protons for atoms. This activity has 37 fill in the blank, 38 matching, and 3 short answer...
Royal Society of Chemistry
Some A-level Reagents
Learning names and formulas can be a daunting task for young chemists, so support their study with interactive puzzles! First, users match each formula with its correct name. Then, individuals use them to complete three logic games.
American Chemical Society
Development of Baking Powder
Did you know baking powder can be used to treat acne, whiten teeth, and make sugar cookies? The lesson on the development of baking powder is ready-to-go with no preparation required. Through readings, pupils answer questions, complete...
Curated OER
Polymers
Students explore online tutorial on polymers. In this chemistry lesson, they create two polymers in the lab and compare their properties. They write a sales letter about their new and improved polymer product.
Curated OER
Covalent Compound Scavenger Hunt
In this chemistry learning exercise, students look for the covalent compounds while examining the objects in the classroom. They also apply the knowledge of formulas.
Curated OER
Bonding Review
Students differentiate metallic, ionic and covalent bonding. In this chemistry instructional activity, students explain how these bonds are formed. They classify substances according their bond type.
Curated OER
Chemical Bonding
Eighth graders identify the three main types of chemical bonds. In this chemistry lesson plan, 8th graders draw Lewis dot diagrams of elements and determine the bond formed. They create a model water molecule.
Curated OER
A Very Simple Conductivity Meter
Students conduct a simple conductivity meter to test the ionic or non-ionic nature of water samples including one from their home.
Virginia Department of Education
Vapor Pressure and Colligative Properties
Hate to vacuum, but enjoy using a vacuum pump? Explore a instructional activity that starts with a demonstration of boiling water at various temperatures by using a vacuum pump. Then scholars design their own experiments to measure vapor...
Virginia Department of Education
Matter and Energy: Equations and Formulas
Using simple materials, an informative instructional activity demonstrates the Law of Conservation of Matter and explains how to balance chemical equations. Young chemists perform experiments, analyze reactions, and balance chemical...
Pingry School
Comparing Activities of Selected Metals
Don't overreact! A simple experiment demonstrates chemical reactions as scholars mix chemical solutions and metals in a large well plate. They note all changes to the metals, solutions, precipitate, colors, and more. A full data table...
Curated OER
Unit 7 Acid and Base
A series of 25 multiple choice questions is presented to chemistry learners to review properties of acids and bases. A page of notes precedes the questions and contains information about the Arrhenius and Bronsted-Lowry definitions,...
Curated OER
Covalent Compounds Worksheet
In this chemistry worksheet, students examine the concept of covalent bonds with the assessment. The answer key is included on page 3 and 4.
Curated OER
The Speed of Chemistry
In this rates of chemical reactions worksheet, students read about the various factors that influence the rate of chemical reactions such as surface area, concentration, temperature and catalysts. Students answer eight questions about...
Curated OER
Electronegativity
In this electronegativity learning exercise, high schoolers complete a chart given 10 molecules. They draw their Lewis structures, they draw the shape of the molecule, they determine the difference in electronegativity between the bonds,...
Curated OER
Solubility and Intermolecular Forces
For this solubility and intermolecular force worksheet, students are given 26 clues about forces between molecules such as hydrogen bonds and London forces and about solubility of solutions.
Curated OER
Unit 4 - Periodic Table
There are 17 multiple choice questions on this periodic table test. The focus is on properties of the elements by group and period. The exam is not formatted well, making it a little difficult to read. There is also a typographical...
Kenan Fellows
What Element Would You Be?
Primo Levi wrote a collection of short stories comparing his life from Italy to Auschwitz to elements in the periodic table. Pupils read an excerpt from his book and research the characteristics of various elements. Then, they make a...
Curated OER
Chemical Reactions
In this reactions worksheet, learners state the differences between a coefficient and subscript and an exothermic and endothermic reaction. Students review the different types of reactions. This worksheet has 13 short answer questions.
Curated OER
Studying with Chemical Compounds
Learners create a foldable to help them remember topics on chemical compounds. In this physical science lesson, students differentiate ionic and covalent compounds. Given certain compounds, they identify whether it's ionic or covalent.
Curated OER
Review Sheet for Covalent Compound Quiz
In this covalent compounds worksheet, students name 6 compounds given their chemical formulas, they write the formulas for 6 compounds given their chemical names and they answer questions about boiling points and solubility. They write...