Hi, what do you want to do?
CK-12 Foundation
Ck 12: Periodic Trends
[Free Registration/Login may be required to access all resource tools.] In the following online tutorail students will use the Periodic Table to identify and explain periodic trends, including atomic and ionic radii, electronegativity,...
CK-12 Foundation
Ck 12: Polarity and Intermolecular Forces
[Free Registration/Login may be required to access all resource tools.] The following online tutorial describes how the electronegativity difference between two atoms in a covalent bond results in the formation of a nonpolar covalent,...
CK-12 Foundation
Ck 12: Polarity in Chemical Bonds
[Free Registration/Login may be required to access all resource tools.] Students will determine the polarity of a chemical bond using the electronegativity chart, and then distinguish between nonpolar covalent, polar covalent, and ionic...
Concord Consortium
Concord Consortium: Stem Resources: Chemical Bonds
By working through this web-based activity, students differentiate between ionic, non-polar covalent, and polar covalent bonds. Specifically, distinctions are made between bonding types based on orbital shapes and electronegativity...
Texas Instruments
Texas Instruments: Setting the Trend
In this activity you will - Determine the trend in atomic radius for the periodic table. Determine the trend in first ionization energy for the periodic table. Determine the trend in electronegativity for the periodic table.
University of Colorado
University of Colorado: Ph Et Interactive Simulations: Molecule Polarity
When is a molecule polar? Change the electronegativity of atoms in a molecule to see how it affects polarity. See how the molecule behaves in an electric field. Change the bond angle to see how shape affects polarity. See how it works...
Texas Education Agency
Texas Gateway: Periodic Table Trends
Given descriptions, scenarios, or groups/series of elements, students will use the Periodic Table to relate the size of the atomic radii, electronegativity, and ionization energy of elements to their position on the chart. Students will...
OpenStax
Open Stax: Andrew R. Barron: Valence Shell Electron Pair Repulsion (Vsepr) Theory
Detailed explanation of Valence Shell Electron Pair Repulsion (VSEPR) Theory with examples and questions for the reader to check comprehension. Also includes step-by-step instructions for correctly building molecules using VSEPR Theory....
Royal Society of Chemistry
Royal Society of Chemistry: Gridlocks: Level 3
A collection of grid puzzles that cover a wide variety of topics in advanced high school chemistry. These are excellent for topic review and reinforcement. The puzzles can be played online and also downloaded as worksheets. Answers are...
Other
University of Texas Dallas: Electron Appetites
The first section of this lecture on polarity and electronegativity discusses what polar covalent bonds are by using carbon dioxide as an example.
University of Arizona
University of Arizona: Biochemistry
Problem sets, tutorials, and activities related to biochemistry.
Simon Fraser University
Chem1 Virtual Textbook: Polar and Nonpolar Bonds
The General Chemistry Virtual Textbook, or Chem 1, is broken into several sections covering various aspects of topics related to chemistry. This section deals with chemical bonding and specifically the study of both polar and nonpolar...
CK-12 Foundation
Ck 12: Trends in the Periodic Table
[Free Registration/Login may be required to access all resource tools.] Students investigate specific properties that can be predicted by an element's position on the periodic table. Additionally, they will look at the formation of ions,...
Concord Consortium
Concord Consortium: What Makes Water Special?
Activity 4 investigates How does electron distribution impact our observations? Students have described how properties of molecules change depending on what atoms make up the molecule. In this activity students will use those ideas to...
University of Southern California
Atomic Bonds
This slide show on atomic bonds contains several slides on electron affinity. Other topics include covalent, Sigma and Pi bonds, and atomic bonding in solids.
Environmental Chemistry
Periodic Table of Elements: Gallium
A very detailed look at the element Gallium, a member of the Boron Group.
Environmental Chemistry
Periodic Table of Elements: Indium
A very detailed look at the element Indium, a member of the Boron Group.
Environmental Chemistry
Periodic Table of Elements: Thallium
A very detailed look at the element Thallium, a member of the Boron Group.
CK-12 Foundation
Ck 12: General Chemistry
[Free Registration/Login may be required to access all resource tools.] Students will be introduced to some of the basic concepts of chemistry.
CK-12 Foundation
Ck 12: Chemistry: Bond Polarity
[Free Registration/Login may be required to access all resource tools.] Covers polarity and covalent bonds.
Concord Consortium
Concord Consortium: Stem Resources: Electrons in Atoms and Molecules
A module with animations, explanations, and questions about the importance of electrons in the structure of an atom. Understand the definition and locations of electrons in the atom. Explore the role of electrons in bonding, polarity,...
Frostburg State University
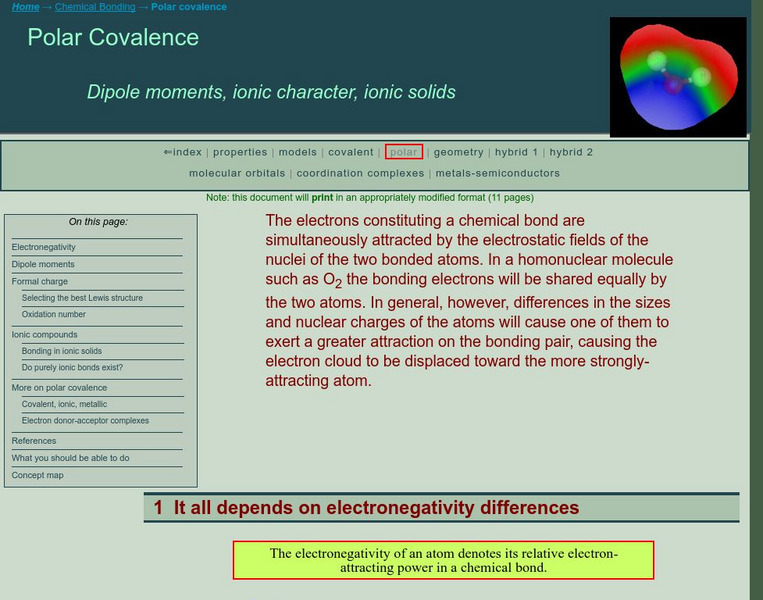
General Chemistry Online: Chemical Bonds Faq
Find answers to common questions about chemical bonds. Recognize molecular orbital theory and know how to draw a Lewis structure.
Educaplus (Jesús Peñas Cano)
Educaplus: Polaridad De Los Enlaces [In Spanish]
Test your knowledge by sorting the links from the lowest to greatest polarity.
Educaplus (Jesús Peñas Cano)
Educaplus: Polaridad Y Diferencias De Electronegatividad [In Spanish]
Test your knowledge of Chemistry. Complete the diagram by dragging the labels to the correct location.
Other popular searches
- Chemistry Electronegative
- 3 D Electronegative
- Trends Electronegative
- Chem 3d Electronegative
- Electronegative Energy
- Electronegative Shopping

















![Educaplus: Polaridad De Los Enlaces [In Spanish] Unknown Type Educaplus: Polaridad De Los Enlaces [In Spanish] Unknown Type](https://content.lessonplanet.com/knovation/original/180051-fd27cf1353791aa3603432e82287e753.jpg?1661774243)
![Educaplus: Polaridad Y Diferencias De Electronegatividad [In Spanish] Activity Educaplus: Polaridad Y Diferencias De Electronegatividad [In Spanish] Activity](https://content.lessonplanet.com/knovation/original/180052-bebdac3e107b5cf58c094c0fe5c0176c.jpg?1661810230)