Hi, what do you want to do?
Khan Academy
Khan Academy: Dalton's Law of Partial Pressure
Use this tutorial to understand Dalton's Law of partial pressure, and then answer some interactive practice questions to review the material.
Chiral Publishing
Chiral Publishing: An Introduction to Chemistry: Dalton's Law of Partial Pressure: Audio Book
This audio book provides a quick review of Dalton's law of partial pressure. The author narrates with the help of a picture, equations, and a description of how to use the equations.
Michael Blaber, PhD
Florida State University: Gases: Gas Mixtures & Partial Pressure
Avogadro's law is stated in equation form. The lesson does not include much discussion, but it does relate the partial pressure and the total pressure of a gas sample to the mole fraction of its components.
Chiral Publishing
Chiral Publishing: An Introduction to Chemistry: Gases [Pdf]
This "Introduction to Chemistry" chapter discusses gases and their properties, ideal gas calculations and equation stoichiometry, and Dalton's law of partial pressures. Many pictures, charts, and sample calculations help to further...
Science Education Resource Center at Carleton College
Serc: Making Hydrogen Gas From a Decomposition Reaction
A very simple reaction of acid (HCl) and metal (magnesium) to produce Hydrogen gas and metal salt will provide the volume of gas to evaluate. Students will determine the amount of gas produced at room conditions and then using the...
Other
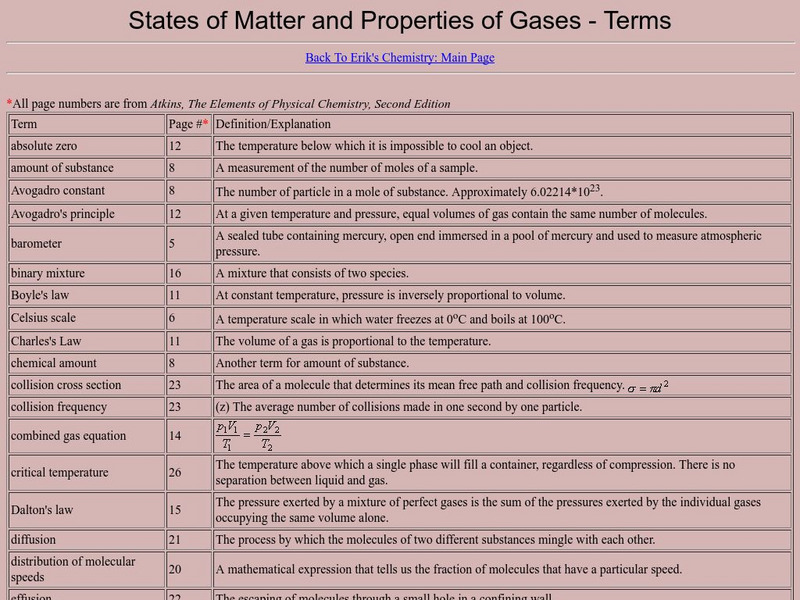
States of Matter and Properties of Gases: Terms
A very complete list of terms that are important to the study of gases. This resource is a web archive.
CK-12 Foundation
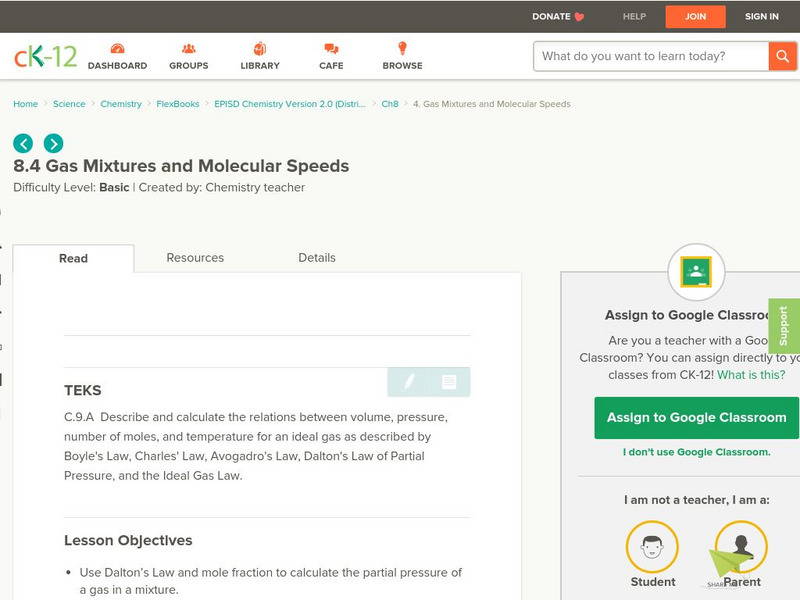
Ck 12: Gas Mixtures and Molecular Speeds
[Free Registration/Login may be required to access all resource tools.] The following online tutorial helps students use Dalton's Law and mole fraction to calculate the partial pressure of a gas in a mixture. They will learn to calculate...
University of Florida
Chemistry 2041 Lecture Notes: Ideal Gases
The ideal gas law is presented and explained. The derivation of other gas laws is performed. Gas behavior is explained in terms of gas laws. Excellent graphics.