Hi, what do you want to do?
Curated OER
Atomic Structure
A flashy PowerPoint includes clear summaries of atomic structure and electronic configuration. Dot and cross diagrams will help viewers learn how to draw an atom. As a result, learners should be more clear how electronic configuration...
Curated OER
Biochemistry Assignment
In this biochemistry worksheet, students complete a table by filling in the missing information about different elements. Students draw the Bohr diagram and the Lewis dot diagram for several atoms.
Curated OER
Metals vs. Non-Metals
In this elements activity, students compare and contrast the characteristics of metals and non-metals. Students practice drawing Lewis dot diagrams and writing ion notation. This activity has 9 word problems, 8 matching questions, and 5...
Curated OER
Ionic Bonding
In this ionic bonding worksheet, students review the characteristics of ionic bonding, draw Lewis dot diagrams for elements, and write the chemical formula for ionic compounds. This worksheet has 8 drawings, 14 fill in the blank, and 6...
Curated OER
How Do Atoms Stick Together?
In this chemical bonding instructional activity, learners answer 76 questions about compounds, Lewis dot structures, intermolecular forces between atoms, electronegativity and bonding and types of bonds.
Curated OER
Thermochemistry
In this thermochemistry worksheet, students indicate whether the given processes are endothermic or exothermic reactions. Students complete the phase change diagram as well as define a given set of vocabulary words. Students calculate...
T. Trimpe
Atomic Basics
Get down to basics with these worksheets on the structure of atoms. Challenging young chemists to identify information from the periodic table and create Bohr diagrams and Lewis dot structures for different elements, this...
National Institute of Open Schooling
Chemical Bonding
Name is Bond, covalent bond. Through readings and answering questions, classes explore the different types of chemical bonds, their characteristics, valence shell electron pair repulsion theory, and atomic orbitals.
Curated OER
2007 U.S. National Chemistry Olympiad Local Section Exam
Sixty multiple choice questions cover the entire gamut of chemistry concepts. This is the local section of the U.S. National Chemistry Olympiad, where your chemistry candidates take a shot at entering the national competition. They...
Curated OER
Oxidation Numbers
In this oxidation numbers worksheet, learners determine the oxidation number for given elements and draw Lewis Dot Diagrams. This worksheet has 6 fill in the blank and 11 short answer questions.
Curated OER
Ionic Compounds
In this ionic compounds worksheet, students identify the number of electrons gained or lost in ions, they show that transfer of electrons in ionic compounds and they write balanced ionic compounds given ions. They answer 4 questions...
Curated OER
Chemical Compounds
For this chemical compounds worksheet, students write the name of compounds from their chemical formula, balance equations, write electronic configurations for elements, and draw Lewis dot structures. This worksheet has 8 fill in the...
Curated OER
VSEPR THEORY: BALLOONS AND MOLECULES
Learners listen to the teacher introduce VSEPR theory and explain molecular geometry. They use balloons to visualize the shape of orbitals. Students create visuals of molecular orbitals with strings and balloons.
Curated OER
Oxidation Numbers
In this oxidation numbers activity, students compare metals and nonmetals, draw Lewis Dot Diagrams, and determine the oxidation number for the given elements. This activity has 1 multiple choice, 5 short answer, and 12 fill in the blank...
Curated OER
Periodic Table Basics 2
In this periodic table worksheet, students create cards for a annotated periodic table then use it to answer 16 short answer and fill in the blank questions.
Curated OER
Pauli's Magical Water
Students predict the shape of molecules using VSEPR theory. In this chemistry activity, students differentiate a polar and nonpolar molecule. They discuss why water's polarity is very important.
Curated OER
Oh - The Bonds of Matter!
Eighth graders identify the types of bonds elements form. In this chemistry lesson, 8th graders represent valence electrons with dot structures. They draw and label different atoms.
Texas Education Agency
Texas Gateway: Covalent Bonding: Electron Dot Diagrams
Given descriptions, diagrams, scenarios, or chemical symbols, students will model covalent bonds using electron dot formula (Lewis structures).
Texas Education Agency
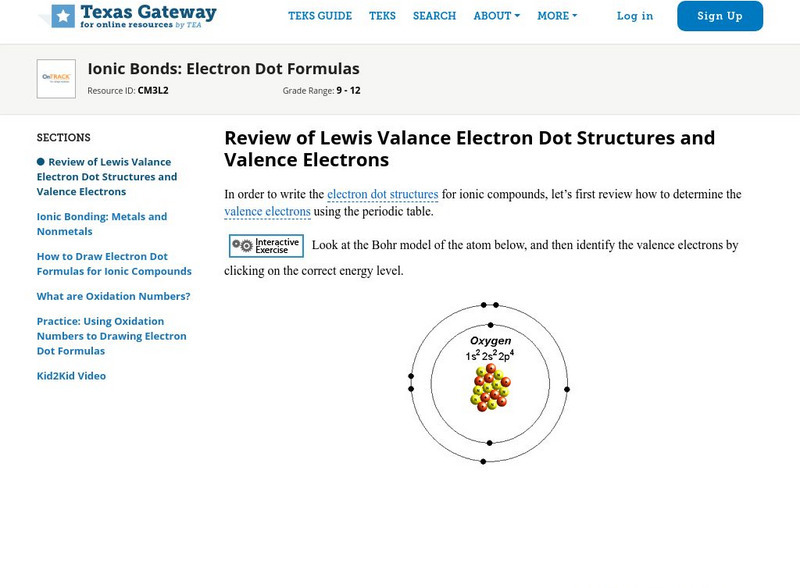
Texas Gateway: Ionic Bonds: Electron Dot Formulas
Given descriptions, diagrams, scenarios, or chemical symbols, students will model ionic bonds using electron dot formulas.
American Chemical Society
Middle School Chemistry: Represent Bonding With Lewis Dot Diagrams
Learners draw and interpret Lewis dot diagrams for individual atoms and both covalent and ionic compounds.
Sophia Learning
Sophia: Lewis Dot Diagrams of Elements: Lesson 4
This lesson will demonstrate how to draw a Lewis Dot diagram for elements. It is 4 of 4 in the series titled "Lewis Dot Diagrams of Elements."
Science Education Resource Center at Carleton College
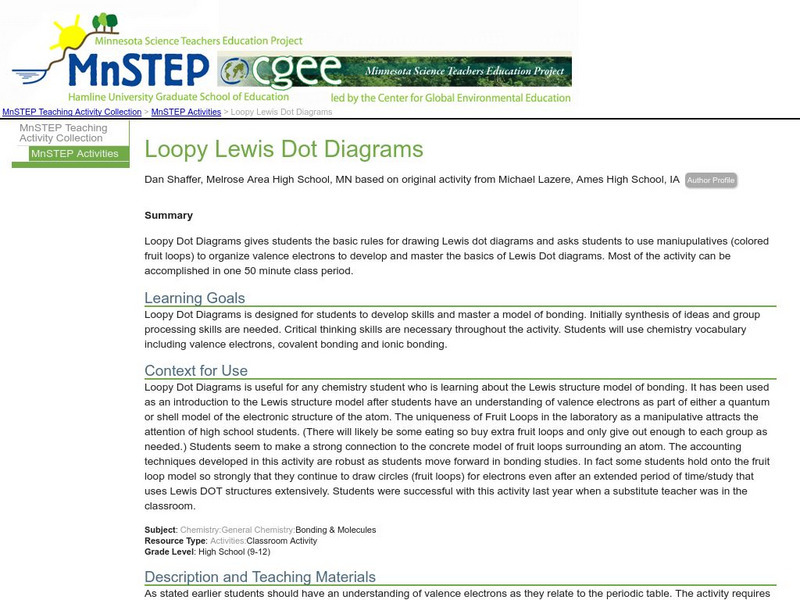
Serc: Loopy Lewis Dot Diagrams
Learners will use colored fruit loops to organize valence electrons to develop and master the basics of Lewis Dot diagrams. They will develop processing and critical thinking skills and also master a model of bonding.
University of California
Organic Chemistry Page: A Brief Tutorial on Drawing Lewis Dot Structures
An excellent lesson on writing Lewis structures. Examples shown include ammonium ion, carbon dioxide and the carbonate ion.
Other
Organic Chemistry: Lewis/kekule Structures
This slide presentation contains a few slides that will be useful for students learning to write Lewis structures. A discussion of multiple bonds is included, and clear examples are shown.