Hi, what do you want to do?
Other
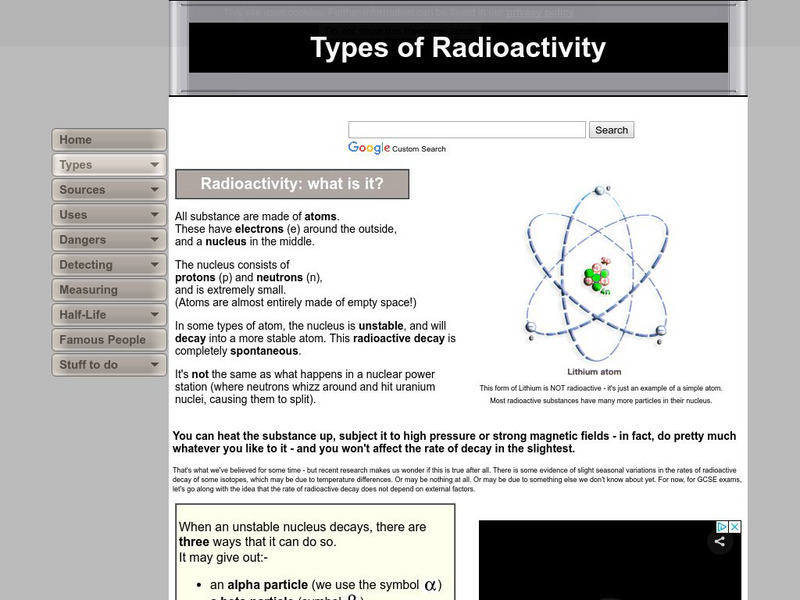
Andy Darvill's Science Site: Types of Radioactivity
Find out about radioactivity, and how it occurs inside an atom's unstable nucleus.
Concord Consortium
Concord Consortium: What Are Nature's Building Blocks?
Activity 3 of this module investigates: How do we know what's inside an atom? From Ernest Rutherford's Gold Foil Experiment to his investigation of the Plum Pudding model, students become aware of the Nuclear Model of an Atom. Also in...
Nobel Media AB
The Nobel Prize: Structure of Matter
This site provides an illustrated and animated story that seeks to answer several questions. What are atoms made of? What is keeping a nucleus intact? What happens if you try to separate two quarks? Are quarks fundamental?
Concord Consortium
The Concord Consortium: Molecular Workbench Zoom It
See examples of scales based on a range from ten to the twelfth meters to ten to the negative twelth meters. Examples range from parts of our solar system to the nucleus of an atom.
American Chemical Society
Middle School Chemistry: Protons, Neutrons, and Electrons
Explore the particles that make up atoms: protons, electrons, and electrons.
CK-12 Foundation
Ck 12: Chemistry: Gold Foil Experiment: Rutherford's Atomic Model
[Free Registration/Login may be required to access all resource tools.] This site explains Rutherford's discovery of the nucleus and the planetary model of the atom and the role of gold foil experiment in refining the atomic model. It...
Other
A World of Particles: The Neutron
A World of Particles offers a history of the discovery of the neutron, an exclusive particle in an atom.
Famous Scientists
Famous Scientists: Aage Bohr
Learn about the life of Aage Niels Bohr, and see how his work was pivotal in the development of the theory of the structure of the atomic nucleus.
BBC
Bbc: Gcse Bitesize: Atomic Structure
This lesson focuses on the structure of atoms. All substances are made from atoms. Each atom is made of a nucleus - containing protons and neutrons - surrounded by electrons. It provides a link to an assessment.
Simon Fraser University
Chem1 Virtual Textbook: Movement of the Electron
Acting as a subtopic of the General Chemistry Virtual Textbook's section on Atoms and the Periodic Table, this site seeks to answer the question, "Why doesn't the electron fall into the nucleus?"
Soft Schools
Soft Schools: Subatomic Particles Quiz
Take this interactive, multiple-choice quiz over subatomic particles, then review your score and any missed questions at the end.
Sophia Learning
Sophia: Subatomic Particles: The Electron: Lesson 3
This lesson will explain that electrons are negatively charged particles with negligible mass and are found in pairs in orbitals surrounding the nucleus of an atom. It is 3 of 3 in the series titled "Subatomic Particles: The Electron."
Sophia Learning
Sophia: Subatomic Particles: The Proton: Lesson 2
This lesson will explain that protons are positively charged particles having a mass of one amu and are found in the nucleus of the atom. It is 2 of 3 in the series titled "Subatomic Particles: The Proton."
Sophia Learning
Sophia: Subatomic Particles: The Neutron: Lesson 2
This lesson will explain that neutrons are particles in the nucleus that have no charge and a mass of one amu. It is 2 of 3 in the series titled "Subatomic Particles: The Neutron."
Wikimedia
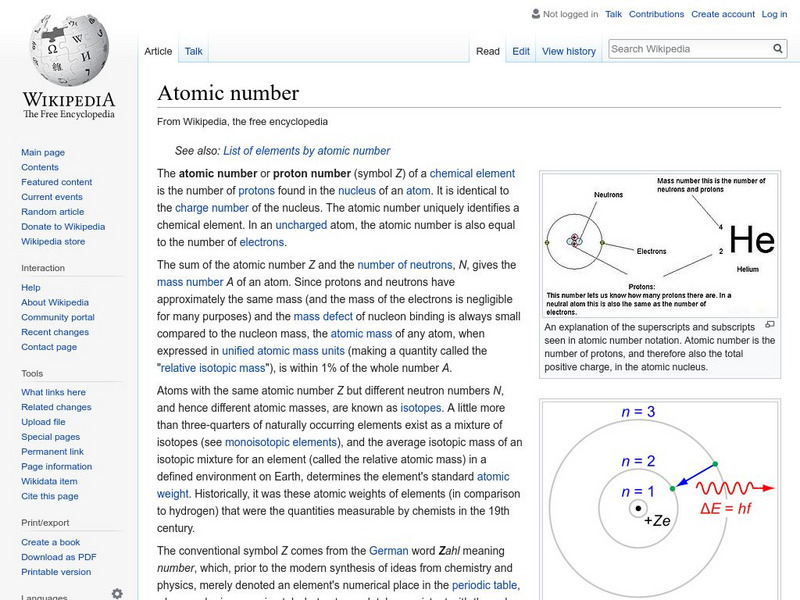
Wikipedia: Atomic Number
Wikipedia provides the definition of the term, "Atomic number," a term used in chemistry and physics to represent the number of protons in the nucleus of an atom.
Physics4kids
Physics 4 Kids: Where Traditional Physics Stops
We're about to move into the modern age of physics. In the early 1800's, scientists began examining the basis of matter, space, and time. Sometimes it gets very confusing, but the big idea is that Newton's physics describe about 90% of...
Energy for Sustainable Development
Esd Bulgaria: Kids & Energy: Uranium (Nuclear)
Nuclear energy is energy in the nucleus of an atom. Atoms are tiny particles that make up every object in the universe. There is enormous energy in the bonds that hold atoms together. Nuclear energy can be used to make electricity. But...
BBC
Bbc: Gcse Bitesize: What Does the Periodic Table Tell Us About the Elements?
The number of protons in the atom of an element determines its place in the Periodic Table. The number of electrons in an atom is the same as the number of protons. These electrons are arranged in shells or 'energy levels' around the...
Science Education Resource Center at Carleton College
Serc: Quantum Atomic Structure
This multi-day lesson plan helps students to understand how the models of the atom have changed and how quantum mechanics affects the electrons as they orbit the nucleus.
BBC
Bbc: Gcse Bitesize: Radiation Treatment
X-rays, gamma rays and beta particles are all used in medicine to treat internal organs. X-rays are produced by firing electrons at a metal target and gamma rays are emitted by the nucleus of radioactive atoms. Gamma rays are used to...
Thomas Jefferson National Accelerator Facility
Jefferson Lab: Coloring Book: Quarks More Than Meets the Eye
Understand and appreciate the research conducted at Jefferson Lab about the smallest particle, the quark.
Purdue University
Purdue University: Fundamental Subatomic Particles
At this site from the Purdue University, the elementary subatomic particles are described and electromagnetic radiation is detailed. Includes learning exercises and answers.
Encyclopedia Britannica
Encyclopedia Britannica: Hydrogen Ion
This brief entry describes the hydrogen ion, strictly, as the nucleus of a hydrogen atom separated from its accompanying electron. The hydrogen nucleus is made up of a particle carrying a unit positive electric charge, called a proton.
Lawrence Berkeley National Laboratory
Berkeley Lab: Basic Nuclear Science Information
Site provides the ABC's of nuclear science including radioactivity and gamma decay to fission and comic rays.