Hi, what do you want to do?
Science Education Resource Center at Carleton College
Serc: How Big Is the Balloon?
A chemistry lab where students investigate limiting reagents and balanced chemical equations. A simple experiment that gives students a physical example of what limiting reactant means.
Alabama Learning Exchange
Alex: S'mores Limiting & Excess Reactants
Students will use manipulatives that represent the ingredients for S'mores (graham crackers, marshmallows, chocolate bar) to determine the limiting and excess reactants (reagents) for making S'mores. The students will extend this to...
Chemistry Collective
Chem Collective: Equilibrium Simulation
This applet is similar to the stoichiometry simulation but applies to chemical reactions that do not go to completion. The calculation of the equilibrium state of such reactions is typically covered months after students learn how to...
Frostburg State University
General Chemistry Online: Limiting Reactant Problem
Explains the essence of limiting reactant problems using a sample problem: How can an amount of product (KNO3) be predicted from amounts for two reactants (KCl and HNO3)? Shows the work and explains how to solve these types of problems.
Chemistry Collective
Chem Collective: Mixed Reception
Participate in the investigation of a virtual crime scene using chemistry concepts to solve a mysterious death. The 40-50 minute activity can be used as a classroom lab or as a homework assignment. Additionally, teachers may request a...
Frostburg State University
General Chemistry Online: The Mole Concept
Resource provides notes on Moles and Stoichiometry. Deals with all book-keeping aspects, including as section on yields and limiting reactants. Includes lesson plans, lecture slides and notes, links to related websites, and frequently...
CK-12 Foundation
Ck 12: Limiting Reactant and Percent Yield
[Free Registration/Login may be required to access all resource tools.] Students will compare theoretical yield to actual yield, and then investigate what happens when one reactant runs out before the other reactants are fully consumed....
Chemistry Collective
Chem Collective: Virtual Lab
Perform virtual lab experiments safely. Pour and mix specific amounts of various solutions on screen and observe changes in temperature and pH. Choose glassware and other tools (ie. bunsen burner, scale) to assist with virtual lab...
Chemistry Collective
Chem Collective: Virtual Lab: Default Virtual Lab Stockroom
Perform virtual lab experiments safely. Pour and mix specific amounts of various solutions on screen and observe changes in temperature and pH. Choose glassware and other tools (ie. bunsen burner, scale) to assist with virtual lab...
Chemistry Collective
Chem Collective: Current Collection of Virtual Lab Problems
A collection of over 30 virtual chemistry problems with accompanying interactive activities. Each problem includes a word file for potential differentiation in your classroom.
Chemistry Collective
Chem Collective: Determining Stoichiometric Coefficients
Students use the virtual lab to determine how four unknown substances react with each other including their stoichiometric coefficients. In this randomized activity, each student receives a different reaction and students can check their...
Chemistry Collective
Chem Collective: Gravimetric Determination of Arsenic
Set in the context of ground water contamination in Bangladesh, this stoichiometry and analytical chemistry activity examines the issues around identifying wells contaminated with arsenic. In this activity, students perform a gravimetric...
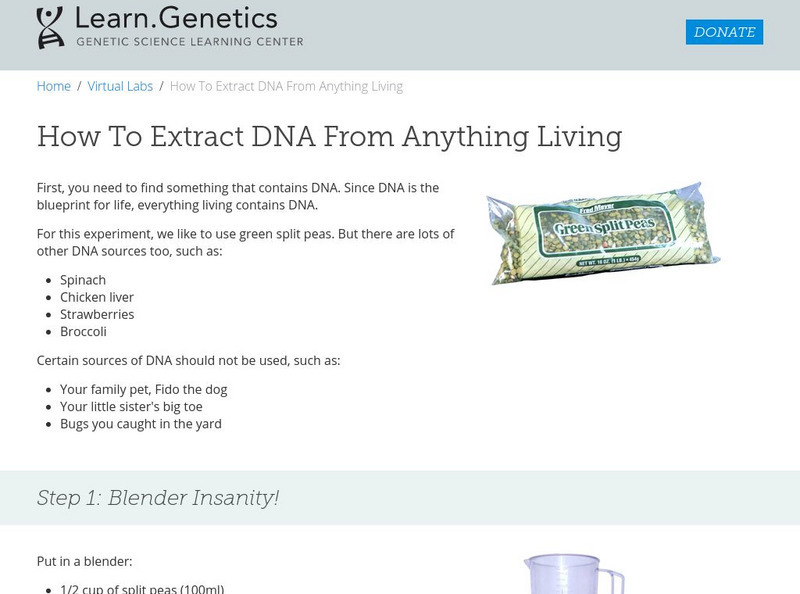
University of Utah
University of Utah: learn.genetics: How to Extract Dna From Anything Living
An experiment to extract DNA from green split peas, spinach, chicken liver, strawberries, and broccoli using simple household reagents.
Sophia Learning
Sophia: Limiting Reactant Theoretical Yield
Find out the procedure in determining the theoretical yield of a product in a chemical reaction. [6:25]
PBS
Pbs Teachers: Fat and Happy?: Testing for Simple Sugars [Pdf]
Investigate different methods for testing the sugar content in foods, and determine relative amounts of sugar in several juices using a sugar indicator solution.
Other
Widener University: Hints for Solving Stoichiometry Prob.
Great analogy if you are uncomfortable with the concept of limiting reactants. A great starting point. Finishes with chemistry.
CK-12 Foundation
Ck 12: Plix Series: Limiting Reactant
[Free Registration/Login Required] Using atoms from a reactants pool, construct products of an equation shown and place them in the product pool. Then answer a challenge question about the topic.
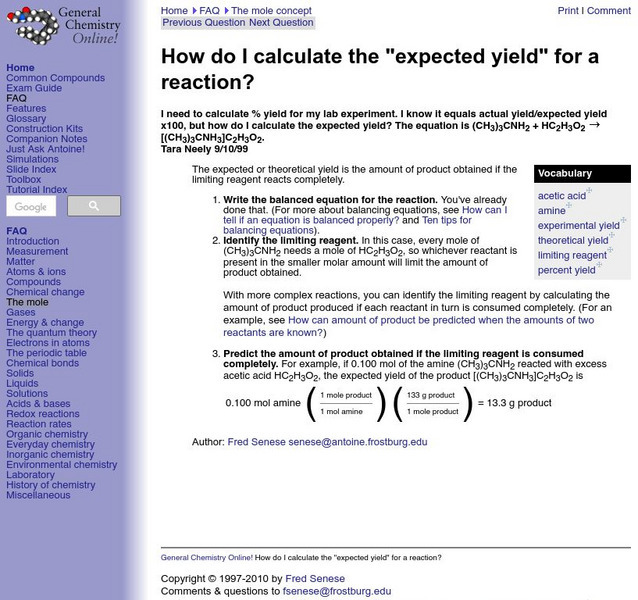
Frostburg State University
General Chemistry Online: Expected Yield
Frostburg State University provides an example of a problem dealing with how to find expected yield. Includes step-by-step directions on how to solve the problem.
Khan Academy
Khan Academy: Hair Dye Chemicals and Cancer
Read the passage Hair Dye Chemicals and Cancer and answer the five-question quiz that follows.
Ducksters
Ducksters: Chemistry for Kids: Chemical Reactions
A site discussing chemical reactions in chemistry including reaction rate, types of reactions, reagents, reactants, catalysts, and inhibitors.
Science Struck
Science Struck: A Simple Guide on How to Calculate Theoretical Yield
Explains vocabulary used when talking about the yield of a chemical reaction and models how to calculate the theoretical and the percentage yield in an example problem.
Chiral Publishing
Chiral Publishing: An Introduction to Chemistry: Chemical Calculations and Chemical Equations [Pdf]
Slide show focusing on the elements of stoichiometry relating to limiting reactants. Go through the first several slides to review or introduce the topic.
Science4Fun
Science4 Fun: Chemical Reactions
What is a chemical reaction? Learn about chemical equations, the four types of chemical reactions, and other fun facts.
Khan Academy
Khan Academy: Gravimetric Analysis and Precipitation Gravimetry
Definition of precipitation gravimetry, and an example of using precipitation gravimetry to determine the purity of a mixture containing two salts.















![Pbs Teachers: Fat and Happy?: Testing for Simple Sugars [Pdf] Activity Pbs Teachers: Fat and Happy?: Testing for Simple Sugars [Pdf] Activity](https://content.lessonplanet.com/knovation/original/67107-890ce2654cdf488a8c3f027b91fd4ae4.jpg?1661419049)