Hi, what do you want to do?
Curated OER
A Solution for Moles
Students calculate the concentration of different solutions. In this chemistry instructional activity, students explain what a solution is. They explain ways to change the concentration of a solution.
Science Buddies
Science Buddies: Saturated Solutions: Measuring Solubility
Many essential chemical reactions and natural biochemical processes occur in liquid solutions, so understanding the chemical properties of liquid solutions is fundamentally important. This project will challenge you to discover how much...
National Association of Geoscience Teachers
Serc: Mentos and Soda Eruptions: Lessons on Explosive Volcanic Eruptions
Students will learn about volcanic eruptions, the scientific method, gas saturation, and bubble nucleation by participating in a popular experiment with Mentos candies and soda.
University of Colorado
University of Colorado: Ph Et Interactive Simulations: Molarity
What determines the concentration of a solution? Learn about the relationships between moles, liters, and molarity by adjusting the amount of solute and solution volume. Change solutes to compare different chemical compounds in water.
Vision Learning
Visionlearning: Biological Molecules: Fats and Proteins
An explanation of the importance of fats and proteins in the human diet. Examples of chemical structures of various fats and proteins are used.
University of Colorado
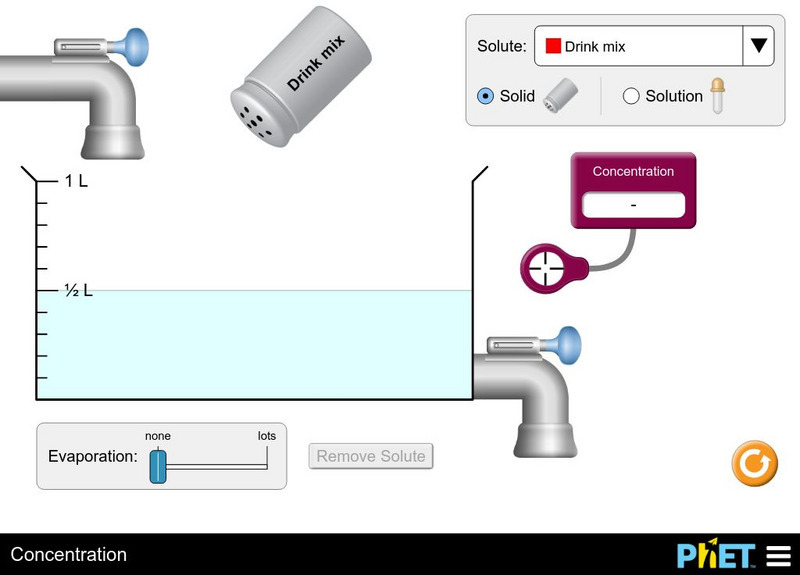
University of Colorado: Ph Et Interactive Simulations: Concentration
Watch your solution change color as you mix chemicals with water. Then check molarity with the concentration meter. What are all the ways you can change the concentration of your solution? Switch solutes to compare different chemicals...