Hi, what do you want to do?
Curated Video
VSEPR Theory: Learn Molecular Geometry Fast - Chemistry Study Guide
Struggling with VSEPR theory and molecular geometry? This video simplifies the concepts you need to master these essential chemistry topics. Learn how to draw Lewis structures with the correct geometry, understand bond angles, and...
Bozeman Science
Lewis Diagrams and VSEPR Models
In this video Paul Andersen explains how you can use Lewis Diagrams and VSEPR Models to make predictions about molecules. The Lewis diagrams are a two-dimensional representations of covalent bonds and the VSEPR models show how the...
Professor Dave Explains
Limitations of VSEPR Theory
We've learned about VSEPR theory, and we know how to use it to predict molecular geometry for a variety of organic molecules. But in fact, there are situations where predictions made with VSEPR theory do not line up with experimental...
Curated Video
VSEPR Theory Postulates: The Rules Behind Molecular Geometry
The Valence Shell Electron Pair Repulsion (VSEPR) theory predicts molecular shapes by focusing on the repulsion between electron pairs surrounding a central atom. The main postulates include: Electron Pair Repulsion: Electron pairs...
Curated Video
VSEPR Theory: Predicting Molecular Shapes with Ease
VSEPR postulates state that the geometry of a molecule depends on the number and arrangement of bonding and lone electron pairs around the central atom
Curated Video
Geometry in BeCl₂ & BF₃: A VSEPR Theory Perspective
BeCl₂ and BF₃ exhibit linear and trigonal planar geometries, respectively, as predicted by the VSEPR theory, minimizing electron pair repulsion
Professor Dave Explains
VSEPR Theory and Molecular Geometry
Did you know that geometry was invented by molecules? It's true! Until the first stars went supernova and littered all the elements across the cosmos, everything was simply spheres, from protons to stars. But then, under cooler planetary...
Professor Dave Explains
Practice Problem: VSEPR Theory and Molecular Geometry
What's with all these shapes? Let's practice assigning hybridization, electron-domain geometry, and molecular geometry. Octahedral! Tetrahedral! Linear! Pyramids and see-saws and what not! Chemistry is fun, isn't it?
Curated Video
VSEPR Theory - lone electron pair influence
Bond Angle Distortion: Lone pairs reduce bond angles between bonding pairs. For example, in a molecule like NH₃ (ammonia), the ideal tetrahedral angle of 109.5° is compressed to around 107° due to the presence of a lone pair on nitrogen....
Professor Dave Explains
Crystal Field Theory
We are used to using a theory like VSEPR theory to predict molecular geometry, but unfortunately with coordination compounds, things are not so simple, because of those pesky d orbitals on the central metal atom. Crystal field theory is...
Professor Dave Explains
Visualizing Molecular Geometry With 3D Software
We've already learned about VSEPR theory, and how to use it to predict the shapes of various molecules. But we didn't cover all of the molecular geometries for certain hybridizations, and we didn't talk about bond angles, so let's do...
Curated Video
Shaping Molecules: Geometry of AB₄, AB₅, and AB₆ Explained
The geometry of molecules with the general formulas AB₄, AB₅, and AB₆ is determined by the Valence Shell Electron Pair Repulsion (VSEPR) theory, which helps predict molecular shapes based on the repulsion between electron pairs around...
Curated Video
Valency Revealed Group IV Elements' Complex Chemistry
Tetrahedron formation is a crucial aspect of the chemical bonding and molecular geometry of Group 4 elements like carbon, silicon, germanium, tin, and lead. The tetrahedral shape is formed by the arrangement of four substituents around...
Catalyst University
General Chemistry | Lewis Structures (Example #3)
In this video, we will determine the Lewis structure for the compound, CHCl3 (chloroform).<br/>
Professor Dave Explains
IIT/JEE Chemistry Practice #17: Molecular Geometry
Practice REAL problems from actual past IIT/JEE exams with Professor Dave!
Professor Dave Explains
IIT/JEE Chemistry Practice #20: Hybridization
Practice REAL problems from actual past IIT/JEE exams with Professor Dave!
Berkeley University of California
VSEPR Examples
Ah is the element of surprise. It is also one of the few elements you won't encounter when doing VSEPR problems. The videos work many examples of VSEPR in various configurations and detail the angle degrees based on the number of atomic...
JFR Science
VSEPR Theory: Determining the 3D Shape of Molecules
Ready to take molecules out of the two dimensional world and into 3-D? Chemistry scholars explore molecular geometry through a well-written video from the JFR Science series. Topics include the effects of bonding and non-bonding...
Berkeley University of California
Molecular Structure and Geometry
While physics is known for the math connections, chemistry is know for geometric connections. The 11th video in the series discusses how to determine the molecular structure of bonded atoms in a 3-D plane using steric numbers.
Khan Academy
Khan Academy: Vsepr for 2 Electron Clouds
An introduction of VSEPR and molecules with 2 electron clouds. [8:04]
Khan Academy
Khan Academy: Vsepr for 3 Electron Clouds
A description of VSEPR with 3 electron clouds and various types of molecules. [12:33]
Khan Academy
Khan Academy: Vsepr for 4 Electron Clouds
VSEPR for molecules with four electron clouds including examples for tetrahedral, trigonal pyramidal, and bent molecules. [10:05]
Khan Academy
Khan Academy: Vsepr for 5 Electron Clouds
Learn about VSEPR for molecules with five electron clouds, and see examples for trigonal bipyramidal and seesaw-shaped molecules. [14:47]
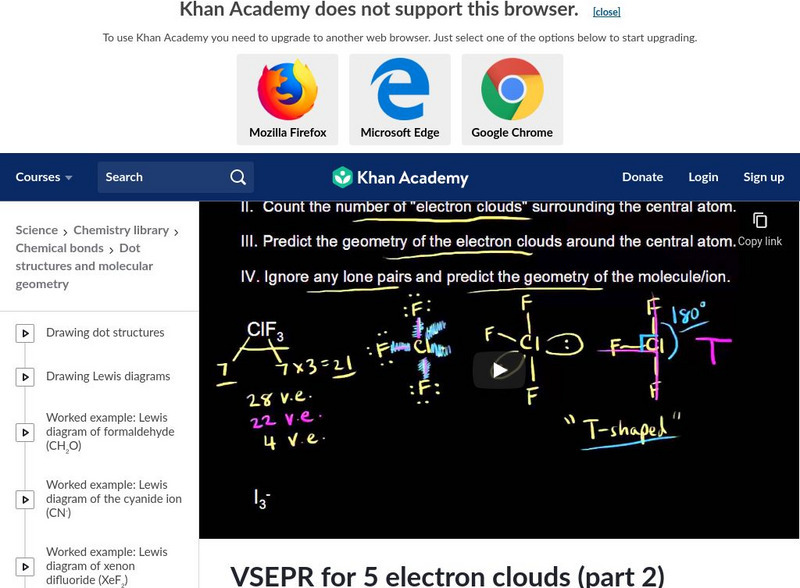
Khan Academy
Khan Academy: Vsepr for 5 Electron Clouds Part 2
Part II of using VSEPR for five electron clouds including examples for T-shaped and linear molecules. [10:48]
Other popular searches
- Vsepr Theory
- Vsepr Models
- Vsepr and Molecular Shapes
- Chemistry Vsepr
- Vsepr Models of Caffiene
- Reactivity and Vsepr Models
- Vsepr Shape Balloons
- Balloons Vsepr