Concord Consortium
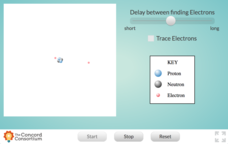
Structure of an Atom
Feeling a little uncertain about your materials for teaching the quantum mechanical model of the atom? Here is an interactive that will help! Chemistry and physics scholars alike will benefit from a simple resource that illustrates the...
Concord Consortium
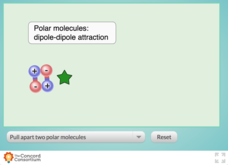
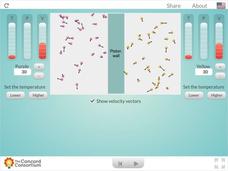
Comparing Dipole-Dipole to London Dispersion
Which intermolecular force is the strongest? Scholars test the relative strength of London dispersion forces, dipole-dipole interactions, and induced dipoles using a simulator. The interactive allows learners to pull on paired molecules...
Concord Consortium
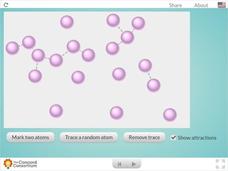
Molecular View of a Gas
Welcome to the fast-paced life of a gas atom! Learners gaze at gaseous atoms up close using a colorful interactive. They can highlight two atoms and observe their movements within their container or follow the dizzying path of one atom...
Concord Consortium
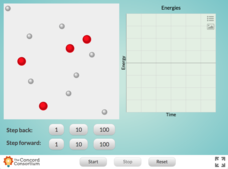
Reaction Between Hydrogen and Oxygen Atoms
Is this resource a great way for your class to observe bonding between oxygen and hydrogen? OH yeah! Scholars learn about the changes in kinetic and potential energy as molecules of oxygen and hydrogen interact. Kinetic, potential, and...
Concord Consortium
Opposites Attract
Whether they pull together or push away from one another, magnets are sure-fire pupil pleasers! Take their study of magnetism to a new level with a fun interactive. Individuals control the polarity of two spheres to observe attractive or...
Concord Consortium
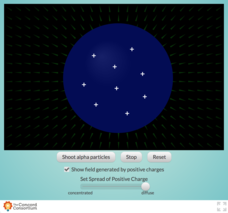
Concentrating Charge and Electric Fields
How did Rutherford determine that the nucleus was the center of an atom? Take a look inside the famous Gold Foil Experiment with an interesting interactive. Learners fire a beam of alpha particles at a nucleus containing variable...
Concord Consortium
Direction of Force Around a Van de Graaff—Negatively Charged
Learn about the direction of magnetic force with a very attractive interactive! Young physicists move a positively charged object around a negatively charged Van de Graaff generator and observe the direction of the forces present. A...
Concord Consortium
Van de Graaff Discharge
Your class will definitely get a charge out of an electric interactive! Show young scientists what really happens when you place a wand near a Van de Graaff generator using a simulation. Pupils add varying amounts of negative charge to...
Concord Consortium
Energy of a Pendulum
Just a swingin'! Introduce physical science scholars to the energy forms associated with a pendulum using a simple interactive. Learners adjust the height from which the pendulum starts, then observe changes in potential, kinetic, and...
Concord Consortium
Collisions and Kinetic Energy
Can your physical science classes describe what happens when two objects collide? Whether they are new to the study of kinetic energy or just brushing up on their skills, pupils can observe the outcome of a variety of collisions using a...
It's About Time
Plate Boundaries and Plate Interactions
How does the Earth continually repair itself? Explore the answer to this question, and others, with a unit on plate boundaries and interaction. Pupils classify the types of movement at plate boundaries and identify the distribution of...
Concord Consortium
Temperature and Reaction Rate
Does increasing temperature increase the rate of a chemical reaction? Junior chemists examine the effects of temperature on reaction rate using an engaging interactive. Pupils select the temperature of the reaction vessel, then observe...
Concord Consortium
Stoichiometry and Balancing Equations
Is your stoichiometry lesson plan just not adding up? Incorporate an exciting interactive to balance things out! Chemistry scholars manipulate the number of molecules added to the reaction vessel, then observe as bonds form and break as...
Concord Consortium
Pressure Equilibrium
All together now! Physical science pupils observe the effects of temperature and amount of substance on pressure and volume of a gas. The interactive resource guides learners through the Combined Gas Law, where they observe changing...
Concord Consortium
Unshared Electrons and the "Bent" Shape
Why is water always so bent out of shape? Scholars investigate the molecular geometry of the water molecule using a 3-D resource. The interactive features options such as rotation and the ability to view electron pairs.
Concord Consortium
Seeing Intermolecular Attractions
Ahh, the rules of attraction...intermolecular attraction! Introduce your chemistry crew to the other forces that influence the behavior of atoms and molecules alike with a simple interactive. Pupils push and pull polar and non-polar...
Concord Consortium
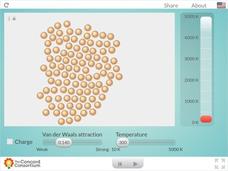
Charged and Neutral Atoms
Do charged and neutral particles behave differently as they undergo phase changes? Science sleuths examine two types of attractive forces using an informative interactive. Pupils can vary the amount of Van der Waals attraction present...
Concord Consortium
Molecular View of a Liquid
Pour on the knowledge with an engaging states of matter interactive! Young physical scientists view the atoms within a liquid to explain what gives a liquid its unique properties. The resource allows users to highlight and observe two...
Concord Consortium
Polymers and Monomers
You don't want to break these chains! Show young chemists and biologists the basics of polymerization with a simple interactive. The resource gives learners three types of polymers to experiment with, as well as a brief tutorial about...
Center for Applied Linguistics
Chemical Interactions: Atoms and Bonding
Watch budding chemists interact with the resource on chemical interactions. In the unit, six lessons provide an overview of basic chemistry, from understanding the development of atomic theory to distinguishing between ionic and covalent...
Colorado State University
How Do Long and Short EM Waves Interact with the Earth's Atmosphere?
Things are about to heat up in your classroom! A kinesthetic lesson asks learners to play the part of the gases in the earth's atmosphere and interact with the sun's radiation. The focus is to learn the impact of the increasing...
Curated OER
Arctica - An Interactive Mystery
Rumours of illness, poisoning, and madness - a ship disappears without a trace! Read this interactive science story and use deductive reasoning skills to solve a mystery. This engaging resource gives science stars a chance to practice...
McGraw Hill
Kepler's Third Law Interactive
Common sense says that the farther a planet is from the sun, the longer the orbit. Kepler turned common sense into his third law by quantifying this relationship. Classes explore Kepler's Third Law through an interactive tutorial. They...
McGraw Hill
Blackbody Radiation Interactive
Noting the color of a star is equivalent to taking its temperature! A creative lesson describes the connection between a star's temperature and the color it emits. As pupils interact with a simulation, they learn how to connect the...