Science Geek
Ionic Compound Formulas
By contrasting cations and anions, this presentation shows how to predict ionic charges by periodic groups. The slides conclude with a few guided practice problems for writing ionic compound formulas.
Virginia Department of Education
A Crystal Lab
Young chemists grow ionic crystals, metallic crystals, and supersaturated crystals in three different lab experiments. Observing these under a microscope allows pupils to compare the various structures.
Curated OER
Ionic vs. Covalent Compounds Lab
In this ionic and covalent compound learning exercise, students investigate the properties of three unknown substances in order to determine if they are ionic or covalent compounds. They write conclusions about their results.
Royal Society of Chemistry
Common Compounds
Can your young chemists identify the most commonly used chemicals in the lab? Introduce the class to the go-to substances in most middle and high school chemistry experiments with an interactive. The resource offers timely feedback as...
Curated OER
Laboratory: Ionic Compound Solubility
For this solubility worksheet, students answer post activity questions about the lab work they completed with ionic compounds. They write sentences about solubility of cations from their lab work. They write chemical reactions for their...
Pingry School
Solubility Product of an Ionic Compound
How do scientists determine when a solution is fully saturated? Scholars address the topic as they observe patterns of precipitation in various concentrations of ions. Using a well plate, pipette, and common chemicals, they collect data...
Curated OER
Types of Bonding Lab
Chemistry lab learners experiment to identify a series of unknown compounds. They choose which tests to perform, but you may suggest melting or boiling point, solubility, electrical conductivity, and malleability. This is a terrific...
Chemistry Collective
Virtual Lab: Textbook Style Limiting Reagents Problems
Time to test the limits! Young scientists make limiting reagent calculations and test their math using a virtual lab simulation. Using their knowledge of ionic compounds, they determine the limiting reagents and the excess compounds.
CK-12 Foundation
Mineral Formation: Evaporating Lake
Get crazy for crystals! Junior geologists learn the secrets of crystal formation through lecture, reading, and examples. Other topics include common ionic compounds found in fresh and salt water, the effects of location on forming...
Carolina Biological Supply
Chemistry Formula Practice
Practice naming chemical compounds or stating the formula when given the name. Discover how well you have mastered these skills either by time or percentage correct.
Curated OER
2001 U.S. National Chemistry Olympiad Part III
Here is a comprehensive method for assessing chemistry learners' knowledge; have them approach two laboratory problems, plan their methods of solving each, and then actually carry out the experiments to find the answers. Both the...
Curated OER
Endothermic and Exothermic Lab
In this endothermic and exothermic reactions worksheet, students experiment with water and a salt of choice to determine if heat is absorbed or released in the reaction. Students take the temperature of their solution over a 2 minute...
Curated OER
Physical Science Review Questions
Prepare your class for a quiz with these physical science review questions. Learners respond to 7 questions about ionic and covalent bonds, oxidation states, and chemical and physical changes. In addition, they name compounds and balance...
Pingry School
Synthesis of an Insoluble Ionic Salt: A Stoichiometry Experiment
Challenge young scientists to design their own experimental procedures. They write the procedure for properly preparing two grams of a water-insoluble ionic salt. To finish, they perform the experiment and collect data to prove their...
Concord Consortium
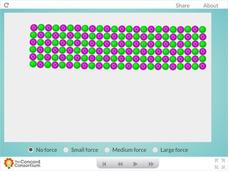
Ceramic Forces
Why are bricks more likely to break than bend? Young science scholars peer inside a ceramic block and examine the effects of downward force at the molecular level. Learners can apply three different levels of force before observing their...
Curated OER
Finding the Formula of Magnesium Oxide
Rather than simply matching up the ionic charges on paper, this exercise gets chemists into the lab to determine the chemical formula for magnesium oxide! If a know amount of magnesium is used, an oxidation reaction results in this...
Curated OER
Precipitate Lab
This lab guide gets chemistry students to determine the identity of five unknown solutions based on reactions with four known solutions. Pupils perform experiments, they write down their observations, they write net ionic equations for...
Curated OER
Solutions and Solubility Review
In this solutions and solubility review worksheet, students are given main ideas about intermolecular forces, concentrations of solutions, molar solutions, ionic equations, solubility rules, acids and bases and titrations. Students...
Curated OER
Chemistry - Science 10
In this chemistry science worksheet, students identify the parts of a chemical reactions. Students name the major particles of the atom. Students apply knowledge about the atom, theories, and models.
Curated OER
Energy and Matter
A review of a full unit on energy and matter, this slide show starts with basic definitions of states of matter and their mass. It then develops the ideas of the forces that that matter can exert. Details about bonding within matter and...
Virginia Department of Education
Matter and Energy: Equations and Formulas
Using simple materials, an informative lesson demonstrates the Law of Conservation of Matter and explains how to balance chemical equations. Young chemists perform experiments, analyze reactions, and balance chemical equations on their...
Curated OER
Solution Chemistry Investigation
Students engage in a instructional activity that is concerned with the concepts related to solution chemistry. They calculate the molar mass of various chemical compounds. Then students predict the anion ratio for ionic compounds....
Curated OER
An Approach to Chemistry via the Analysis of Art Objects: The Scientific Method, Laboratory Safety, Light and Color Theory
Students create a painting that clearly exemplifies the use of primary pigments to make secondary pigments. They demonstrate the distinction between value and saturation. They explain the affect of adjacent colors on each other and...
Santa Monica College
Single and Double Displacement Reactions
If you aren't part of the solution, you are part of the precipitate! Young chemists learn about single and double displacement reactions including precipitation reactions, neutralization reactions, and gas forming reactions. They perform...