Curated OER
Review of Ionic and Covalent Compounds and Transitioning from Ionic to Covalent Compounds
Here is a unique assigment: compare and contrast ionic and covalent compounds in an extensive data table and then analyze Lewis dot structures in antoher. Three columns are to befilled in: "characteristic or feature," "applies to ionic...
Curated OER
Bonding
Almost a complete lesson in itself, this presentation will enhance any bonding and compounds unit. The concepts included are charges of atoms lending to particular molecular structures, the stability of bonds and how to diagram them....
Spark Notes
Review of Chemical Bonding: Review Test
This is an online exercise in which chemistry learners answer a series of multiple choice questions about bonding. Topics addressed include ionic and covalent bonds, electronegativity, ions, valence electrons, resonance structure, and...
Curated OER
Chapter 6 - Bonds
Although there are only 16 questions here, this chemistry handout makes a terrific unit assessment. It queries youngsters on the properties of ionic and covalent compounds, relates bond length tho stability and enrgy, compares polar and...
Curated OER
WS 7.1.3 Molecular and Ionic Compounds
In this compounds activity, students name ionic and molecular compounds given their formulas. They also write the formulas for molecular and ionic compounds given their names.
Curated OER
Bonding
All different types of bonding are covered in this PowerPoint, along with details of resulting bond and molecule shapes. The definitions of traditional molecule shapes and characteristics of behavior are very useful to assist in...
Curated OER
Ranking Molecular Properties Based Upon Intermolecular Forces
A splendid chart of information about chemical bonding tops the first page. In it you will find information on London forces, permanent dipoles, hydrogen bonds, ionic bonds, and covalent bonds. A four-step process for assigning...
Curated OER
Chapter 12 Worksheet - Ions, Ionic Bonding, anc Covalent Bonding
Four pages provide plenty of problem solving practice for chemistry whizzes. They answer questions and write electron configurations for ions. They use Lewis dot diagrams to display equations. Covalent bonds are explored. The last half...
Curated OER
Chemistry of Life Study Guide
Biology learners should understand a few chemistry basics. Here is a worksheet that introduces them to the concepts of chemical reactions, molecular bonding, and the unique combination of properties that make water vital to life....
Curated OER
Ionic bonding
Students explore ionic bonding. They draw examples of ionic bonding and explain the activities of the electrons of the elements. Students use paper plates and candy to draw electron configurations of given atoms.
National Institute of Open Schooling
Chemical Bonding
Name is Bond, covalent bond. Through readings and answering questions, classes explore the different types of chemical bonds, their characteristics, valence shell electron pair repulsion theory, and atomic orbitals.
Curated OER
Atomic Structure and Ionic Bonding (A Visual Approach)
Using toothpicks, marshmallows, and round colored sticky dots, physical science enthusiasts build models of an atomic nucleus. In this eighth grade chemistry lesson plan, they play an atom-naming game with the models that they have...
Curated OER
Bonding
In this word search learning exercise, students locate 12 words related to molecular bonding. Words include molecule, ions, forces, polarity, partial, and particles.
Curated OER
Worksheet-Bonding
In this bonding and electron configuration worksheet, students answer 9 multi-part questions including identifying the differences between bond types, drawing Lewis structures for atoms and identifying types of bonds in molecules.
Curated OER
2007 U.S. National Chemistry Olympiad Local Section Exam
Sixty multiple choice questions cover the entire gamut of chemistry concepts. This is the local section of the U.S. National Chemistry Olympiad, where your chemistry candidates take a shot at entering the national competition. They...
Curated OER
2009 U.S. National Chemistry Olympiad National Exam - Part I
The 2009 version of the first part of a national chemistry competition is posted for your use with olympiad hopefuls. Test takers deal with 60 multiple choice questions covering an entire year of chemistry curriculum. Use this to...
Curated OER
Ionic Compounds
In this compounds worksheet, students review the structure and properties of ionic compounds, their formulas and nomenclature, and molecular mass and percentage composition. This worksheet has 4 short answer questions and 7 problems to...
Curated OER
Chemical Context of Life & Water
Some basic chemistry concepts are fundamental to understanding biology. Learners explore how molecular structure plays a role in biological processes, especially the structure of the water molecule. The final page focuses on water's...
Curated OER
Molecular Models
In this molecular formula worksheet students fill in a table describing elements and chemical formulas. They identify, elements by type and how they bond. The student analyze molecular formulas to identify the number of atoms and if the...
LABScI
Atomic Structure and the Periodic Table of Elements: The Secret Agent Lab
Food always gets attention! Model atomic structure using fruit loops to represent the subatomic particles. After building models, scholars create ionic bonds using their models. Finally, they use these concepts to create a periodic table.
Curated OER
Solubility of Ionic Substances
In this solubility worksheet, students identify substances as either ionic or molecular, define saturated solution and solubility, and calculate concentration of solutions. This worksheet has 7 short answer, 5 fill in the blank, and 27...
Curated OER
Worksheet 7 - A Practice Quiz 3
Every sort of chemical bond is touched upon during this assignment. Chemistry whizzes identify what type of bond is formed by analyzing chemical formulas or Lewis structure diagrams. Multiple choice questions also ask learners about...
Curated OER
Lattice Energy
A few basic facts about lattice energy are listed at the top of the page, including the formula used for calculations. Two charts follow: one for lattice energy and another for ion sizes. There is no work to do on this handout, but it...
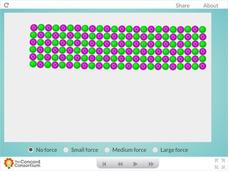
Concord Consortium
Ceramic Forces
Why are bricks more likely to break than bend? Young science scholars peer inside a ceramic block and examine the effects of downward force at the molecular level. Learners can apply three different levels of force before observing their...