Hi, what do you want to do?
Curated OER
Isaac Newton, Part 2/2
The conclusion of a two-part series looks at Isaac NewtonÕs Year of Miracles through a comic lens, making his Law of Universal Gravitation easily understood for those studying action, reaction, inertia, and force.
Bozeman Science
Bozeman Science: Newton's Third Law
Paul Andersen explains how Newton's Third Law applies to all objects. [6:01]
Sophia Learning
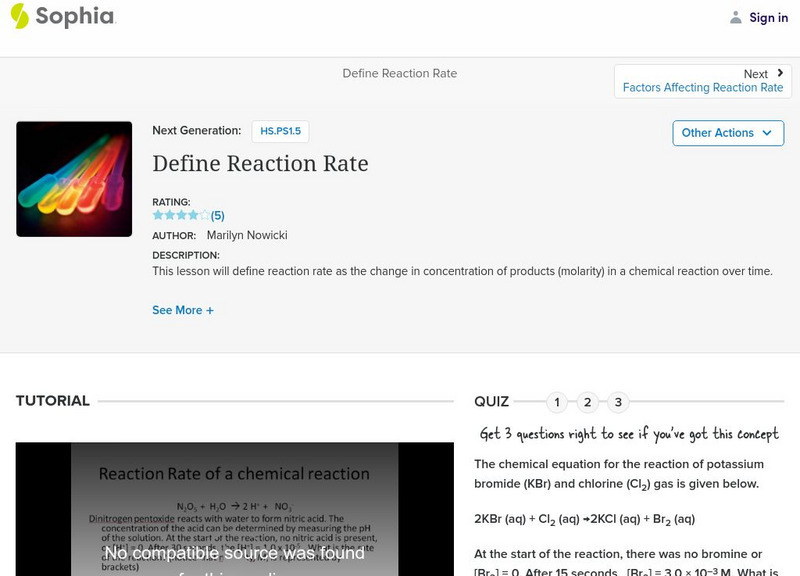
Sophia: Define Reaction Rate
This lesson will define reaction rate as the change in concentration of products (molarity) in a chemical reaction over time.
Sophia Learning
Sophia: Quelling the Adrenaline Response: Lesson 1
At the end of this tutorial, the learner will understand and be able to use basic techniques to reduce intensity of their own fight or flight reaction. It is 1 of 3 in the series titled "Quelling the Adrenaline Response."
Bozeman Science
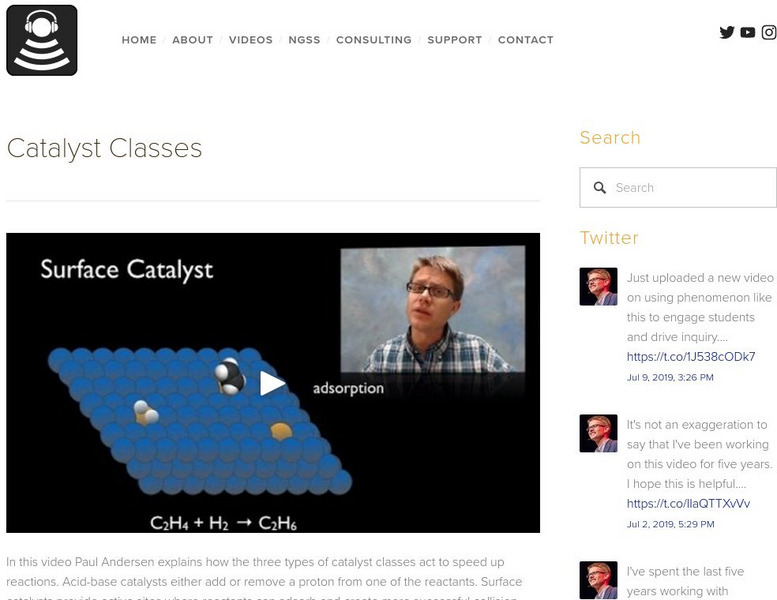
Bozeman Science: Catalyst Classes
In this video Paul Andersen explains how the three types of catalyst classes act to speed up reactions. Acid-base catalysts either add or remove a proton from one of the reactants. Surface catalysts provide active sites where reactants...
Bozeman Science
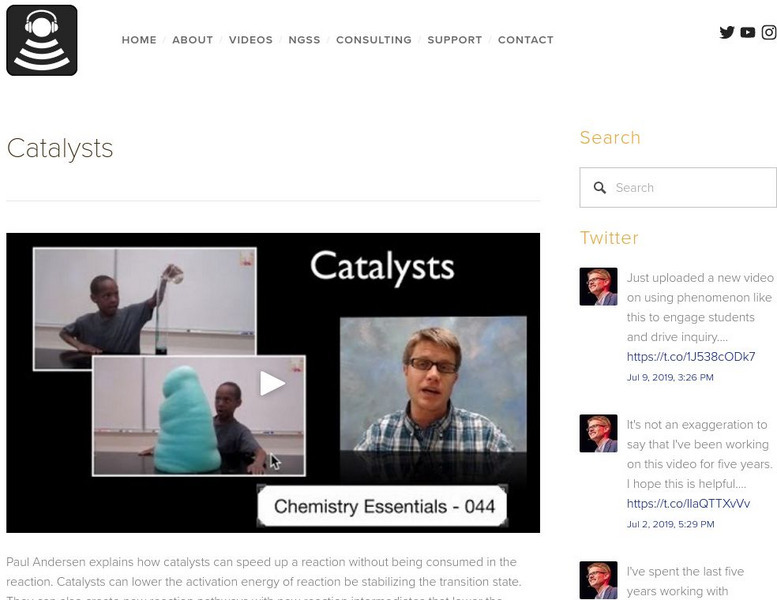
Bozeman Science: Catalysts
Paul Andersen explains how catalysts can speed up a reaction without being consumed in the reaction. Catalysts can lower the activation energy of reaction be stabilizing the transition state. They can also create new reaction pathways...
Bozeman Science
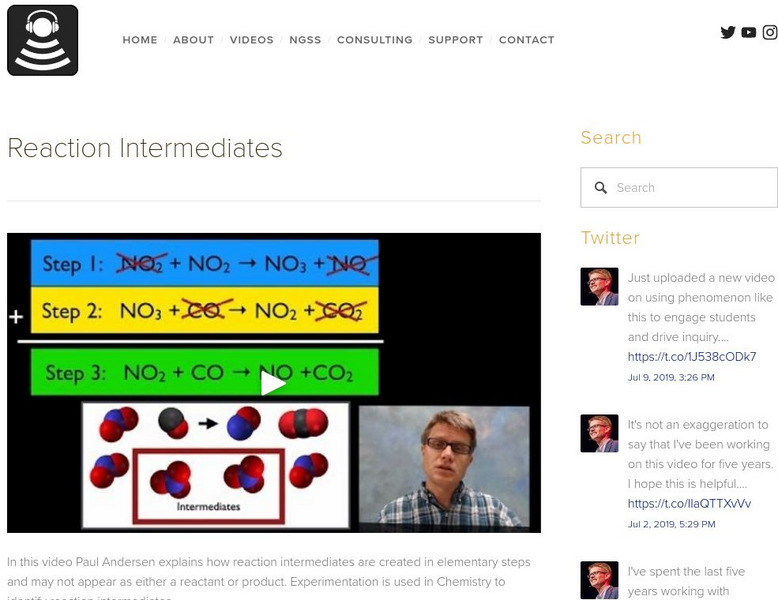
Bozeman Science: Reaction Intermediates
In this video Paul Andersen explains how reaction intermediates are created in elementary steps and may not appear as either a reactant or product. Experimentation is used in Chemistry to identify reaction intermediates. Look at the...
Sophia Learning
Sophia: Chemical Equation: Vocabulary: Lesson 1
This lesson will explain the general format of a chemical equation including products, reactants, arrow notation, and states of matter. It is 1 of 2 in the series titled "Chemical Equation: Vocabulary."
Sophia Learning
Sophia: Chemical Reactions: Summary: Lesson 2
This lesson will discuss how chemical reactions occur and that not all of them will go through to completion. It is 2 of 2 in the series titled "Chemical Reactions: Summary."
Crash Course
Crash Course Physics #5: Newton's Laws
Find out about Newton's Laws of Motion, and the ideas that have developed the foundation of those laws. [11:05]
Khan Academy
Khan Academy: Birch Reduction Ii
The effect of electron-withdrawing and electron-donating groups.
Khan Academy
Khan Academy: Standard Change in Free Energy and the Equilibrium Constant
The relationship between standard Gibbs free energy change and the equilibrium constant K. Calculating K when you know the standard free energy of reaction. [10:45]
Khan Academy
Khan Academy: Hydrohalogenation of Alkynes
See the reaction of an alkyne with a hydrogen halide, leading to formation of a halogenated alkene or alkane. [10:08]
Khan Academy
Khan Academy: Halogenation
Halogenation is a reaction that occurs when one or more halogens are added to a substance. Halogens comprise the seventh column in the periodic table and include fluorine, chlorine, bromine, iodine, and astatine. The resulting product of...
Khan Academy
Khan Academy: Markovnikov's Rule and Carbocations
Learn about Markovnikov's Rule and Carbocations. Figure out which addition reaction is more likely. [9:09]