Hi, what do you want to do?
NOAA
Why Should I Care?: Show How Increased Carbon Dioxide Makes the Ocean More Acidic
How does a change in pH affect the ocean ecosystem? Scholars explore the idea by making an acid-base indicator in part seven of the 10-installment Discover Your Changing World series. First, they explore impacts of carbon dioxide in...
DiscoverE
Ocean Acidification
Combat ocean acidification with bubbles. Young engineers create a system that reduces the acidity of water. Dry ice in water helps simulate ocean acidity, and blowing bubbles into the water results in a gas exchange that neutralizes the...
Teach Engineering
Red Cabbage Chemistry
Using the natural pH indicator of red cabbage juice, groups determine the pH of different everyday liquids. As they work, pupils gain an understanding of pH that may help deal with contaminants in the water supply.
It's About Time
Chemical Names and Formulas
Abracadabra! Provide your class with the tools to perform a chemical "magic show" as they predict the charges of various ions, determine ionic compound formulas, and make observations to determine when a chemical reaction between...
Science Bob Pflugfelder
Science Bob: The Exploding Lunch Bag
This site contains a simple procedure for observing an acid-base neutralization reaction (baking soda and vinegar), in which the product, carbon dioxide, causes a zipped bag to explode.
TeachEngineering
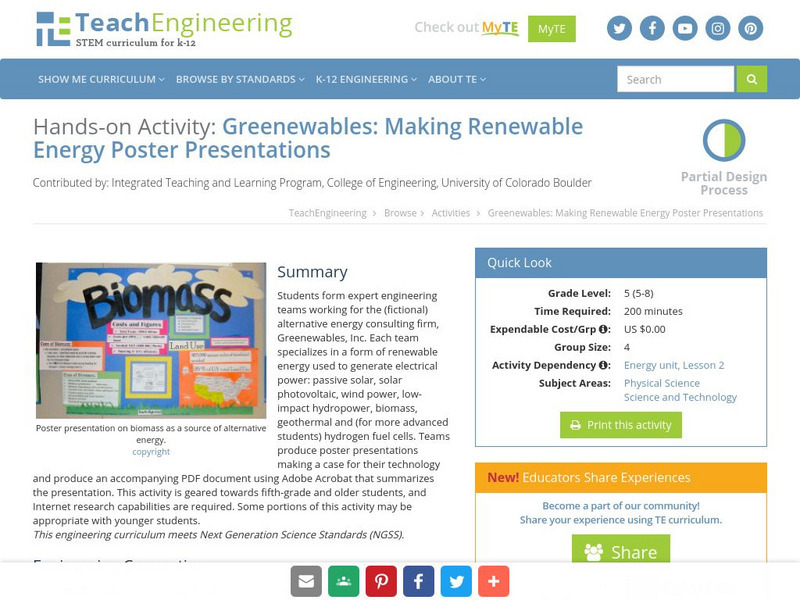
Teach Engineering: Greenewables
Students form expert engineering teams working for the (fictional) alternative energy consulting firm, Greenewables, Inc. Each team specializes in a form of renewable energy used to generate electrical power: passive solar, solar...